DIETARY OLEANOLIC ACID ON THE GROWTH PERFORMANCE, SERUM BIOCHEMICAL, ANTIOXIDANT CAPACITY, AND IMMUNITY OF GRASS CARP FED HIGH COTTONSEED AND RAPESEED MEAL DIETS
-
摘要:
试验旨在研究高比例的棉粕和菜粕饲料中添加齐墩果酸对草鱼生长、血清生化指标、抗氧化和免疫能力的影响。试验选取均重约为50 g的草鱼480尾, 随机分为3组, 每组4个重复, 每个重复40尾。分别为对照组、高比例的棉粕和菜粕组、高比例的棉粕和菜粕组+500 mg/kg齐墩果酸组, 分别记为CON、S1和SQ2。养殖周期为 56d。结果显示: (1)与CON组相比, S1和SQ2组草鱼的末均重(FBW)、增重率(WGR)和特定生长率(SGR)均显著降低(P<0.05), S1组草鱼的饵料系数(FCR)显著升高(P<0.05)。与S1组相比, SQ2组草鱼的FBW、WGR和SGR均显著升高(P<0.05), 而FCR显著降低(P<0.05)。(2)与CON组相比, S1和SQ2组草鱼的血清谷草转氨酶(GOT)、胆汁酸(TBA)、总胆固醇(T-CHO)和肝脏丙二醛(MDA)水平均显著升高(P<0.05)。而血清酸性磷酸酶(ACP)、免疫球蛋白M(IgM)、肠道淀粉酶和肝脏过氧化氢酶(CAT)、超氧化物歧化酶(SOD)、还原型谷胱甘肽(GSH)的水平均显著降低(P<0.05); S1组草鱼的血清低密度脂蛋白胆固醇(LDL-C)的含量显著升高(P<0.05), 而血清C3、肠道脂肪酶和肝脏总抗氧化能力(T-AOC)的含量显著降低(P<0.05)。与S1组相比, SQ2组草鱼的血清GOT、T-CHO和肝脏MDA的水平均显著降低(P<0.05), 而血清ACP、IgM、肠道脂肪酶、淀粉酶和肝脏的CAT、SOD、T-AOC的水平均显著升高(P<0.05)。(3)与CON组相比, S1组和SQ2组草鱼肝脏中gpx1、cat、tgf-β1和il-15的表达水平显著下调(P<0.05); S1组草鱼肝脏中Mnsod和nrf2的表达水平显著下调(P<0.05), 而keap1、tnf-α、nf-κb和il-12β的表达水平显著上调(P<0.05)。与S1组相比, SQ2组草鱼肝脏中Mnsod、gpx1、cat、nrf2、tgf-β1和il-15的表达水平显著上调(P<0.05), 而keap1、tnf-α、nf-κb和il-12β的表达水平显著下调(P<0.05)。综上所述, 高比例棉菜粕饲料显著降低了草鱼的生长性能, 抗氧化能力和免疫力, 并引起草鱼肝脏组织结构的损伤。齐墩果酸的添加可以缓解草鱼肝脏的氧化损伤和炎症反应, 并提高草鱼的生长性能, 抗氧化能力和免疫力。
Abstract:The aim of this experiment was to investigate the effects of oleanolic acid supplementation in high cotton rapeseed meal feed on the growth, serum biochemistry, antioxidant, and immunity of grass carp (Ctenopharyngodon idella). Four hundred and eighty grass carp weighing about 50 g were selected and randomly divided into three groups with four replicates of 40 fish each. These groups included the control group, the high cotton rapeseed meal group, and the high cotton rapeseed meal group supplemented with 500 mg/kg oleanolic acid, which were labeled as CON, S1, and SQ2, respectively, with a culture period of 56d. The results showed that: (1) Grass carp in the S1 and SQ2 groups exhibited significantly lower FBW, WGR, and SGR compared with the CON group (P<0.05), with the S1 group showing significantly higher FCR (P<0.05). (2) FBW, WGR, and SGR were significantly higher (P<0.05), and FCR was significantly lower (P<0.05) in grass carp in the SQ2 group compared to the S1 group. Additionally, compared to the CON group, serum GOT, TBA, T-CHO, and hepatic MDA levels were significantly higher (P<0.05) in the S1 and SQ2 groups. Conversely, levels of serum ACP, IgM, and hepatic CAT, SOD, and GSH were significantly decreased (P<0.05). Furthermore, serum LDL-C levels were significantly higher (P<0.05) in the S1 group, whereas serum complement C3 and liver T-AOC levels were significantly lower (P<0.05). Compared to the S1 group, the SQ2 group exhibited significantly lower levels of serum GOT, T-CHO, and hepatic MDA (P<0.05), and significantly higher levels of serum ACP, IgM, and hepatic CAT, SOD, and T-AOC (P<0.05). (3) Compared to the CON group, hepatic expression levels of gpx1, cat, tgf-β1, and il-15 were significantly down-regulated (P<0.05). Moreover, in the livers of grass carp in S1 group, expression levels of Mnsod and nrf2 were significantly down-regulated (P<0.05), whereas the expression levels of keap1, tnf-α, nrf-κb. and il-12β were significantly up-regulated (P<0.05). Conversely, expression levels of Mnsod, gpx1, cat, nrf2, tgf-β1, and il-15 were significantly up-regulated (P<0.05), while the expression levels of keap1, tnf-α, nf-κb, and il-12β were significantly down-regulated (P<0.05) in the livers of the grass carp in the SQ2 group compared to the S1 group. In conclusion, the high percentage of cotton rapeseed meal in the feed significantly reduced the growth performance, antioxidant capacity, and immunity of grass carp, and induced damage to the liver tissue structure. However, supplementation with oleanolic acid alleviated oxidative damage and inflammatory responses in the liver of grass carp, while improving growth performance, antioxidant capacity, and immunity.
-
Keywords:
- Oleanolic acid /
- Antioxidant indexes /
- Immunity /
- Ctenopharyngodon idella
-
长期以来, 鱼粉和豆粕作为水产饲料首选的优质蛋白质来源, 在水产饲料行业供不应求[1]。但随着集约化养殖和规模化养殖的快速发展, 其限制行业可持续发展的问题逐渐凸显, 如饲料成本上升、供应不稳定等, 因此使用棉粕、菜粕等廉价植物蛋白替代鱼粉已经成为热点[2]。棉籽粕和菜籽粕由于蛋白质含量高, 价格低廉, 来源丰富, 被广泛用于水产饲料当中。然而, 诸如棉籽粕和菜籽粕等植物蛋白源均含有难以消解的抗营养因子, 如棉酚、单宁、芥子碱和硫代葡萄糖苷等[3, 4], 这些抗营养因子可能会对水生动物的机体造成氧化损伤和破坏。研究表明, 在欧洲鲈(Dicentrarchus labrax)[5]、草鱼(Ctenopharyngodon idella)[6, 7]、大菱鲆(Psetta maxima L.)[8]、红鲷(Pagrus major)[9]上均发现饲料中高比例的棉籽粕和菜籽粕都会对生长性能、抗氧化能力和非特异性免疫反应产生不利影响。因此, 缓解植物蛋白对水产动物造成的损伤是当前水产饲料研究的热点, 当前常采用的是发酵、酶解、使用功能性添加剂等措施[10—12]。相对于发酵和酶解的不稳定和标准不一, 功能性添加剂的使用凸显一定的优势, 研究表明使用功能性添加剂可以减轻由高比例的棉粕和菜粕饲料引起的水产动物生长性能降低和生理功能受损等问题[12]。
与其他功能性添加剂相比, 中草药提取物由于其副作用小、毒性低、高效和低成本的优势, 已成为水产饲料研究的重要方向[13]。其中齐墩果酸(Oleanolic acid)属五环三萜类化合物, 主要由女贞子、迷迭香等植物提取而来[14]。并且齐墩果酸被动物机体吸收后具有保护肝脏、降低血脂、提高抗氧化能力、免疫力及抗炎等功效[15—17]。
草鱼是我国养殖规模和产量最大的鱼类, 具有巨大的经济价值[18]。然而, 齐墩果酸在水产养殖业中的应用较少。本课题组前期在黄鳝(Monopterus albus)、中华鳖(Pelodiscus sinensis)和克氏原螯虾(Procambarus clarkii)[19]中发现, 饲料中齐墩果酸的最适添加量为500 mg/kg。因此, 本实验通过在高比例的棉粕和菜粕饲料中添加齐墩果酸, 来探讨对草鱼生长性能、抗氧化能力和非特异性免疫的影响。为齐墩果酸在水产养殖中的应用提供理论基础。
1. 材料与方法
1.1 实验材料
齐墩果酸由湖南植之源农牧有限公司所提供。根据草鱼的营养标准制作饲料, 以鱼粉为主要动物蛋白源, 豆粕、棉粕和菜粕为植物蛋白源, 豆油为脂肪源, 面粉为糖源, 实验共配制3组等氮等脂的饲料(表 1), 分别为CON (基础日粮组)、S1 (高比例的棉粕和菜粕日粮组)、SQ2 (在S1组中添加500 mg/kg的齐墩果酸)。实验所有饲料原料粉碎过60目筛后, 将各饲料原料充分混合均匀, 再加入一定比例的豆油和蒸馏水在V型搅拌机中充分混匀, 最后利用膨化机挤压制成直径为2.0 mm的膨化饲料。
表 1 饲料组成和营养水平(干物质基础, %)Table 1. Composition and nutritional level of diets (DM basis, %)原料Ingredient 组别Group CON S1 SQ2 鱼粉Fish meal 2.00 0 0 豆粕Soybean meal 27.00 0 0 棉粕Cottonseed meal 12.00 28.00 28.00 菜粕Rapeseed meal 12.00 27.00 27.00 面粉Wheat flour 20.00 20.00 20.00 米糠Rice bran 10.00 10.00 10.00 麦麸Wheat bran 3.76 1.36 1.31 豆油Soybean oil 2.50 2.90 2.90 干酒糟及其可溶物DDGS 8.00 8.00 8.00 胆碱Choline 0.20 0.20 0.20 磷酸二氢钙Ca(H2PO4)2 1.50 1.50 1.50 预混料Premixa 1.00 1.00 1.00 防霉剂Antimold 0.03 0.03 0.03 抗氧化剂Antioxidants 0.01 0.01 0.01 齐墩果酸Oleanolic acid 0 0 0.05 合计Total 100.00 100.00 100.00 营养成分Nutrient levelsb 粗蛋白Crude protein 31.22 31.12 31.14 粗脂肪Crude lipid 6.78 6.64 6.81 粗灰分Crude ash 7.20 7.28 7.27 游离棉酚Free gossypol (mg/kg) 99.81 233.52 231.17 注: a预混料来源于青岛玛斯特生物技术有限公司, 成分参考本课题组前期研究[19]; b营养组成为测定值Note: aPremixes are obtained from Qingdao Mast Bio-technology Co., Ltd. and the ingredients are referred to the previous research of this group[19]; bProximate composition are measured values 1.2 饲养管理
草鱼养殖试验在常德西洞庭实验基地的网箱(2.0 m×2.0 m×1.6 m)中进行, 苗种购自西洞庭某农场。试验草鱼先驯化暂养2周后进行正式试验。选取规格整齐、健康、均重约50 g左右的草鱼, 共480尾。试验共3个组, 每个组4个网箱。实验期间每日投喂饲料3次(08:00、12:00和17:00), 且根据天气和摄食情况按照体重的3%—5%进行调节。养殖期8周。养殖期间平均水温为(31.20±4.65)℃、氨氮含量小于0.05 mg/L、亚硝酸盐含量小于0.01 mg/L、pH为8.2±0.2。
1.3 样品采集
在养殖结束后, 草鱼停食24h。记录每个网箱草鱼数量并称总重, 用于统计存活率、饲料系数、增重率。在每个重复中随机取5尾草鱼用丁香油麻醉后, 使用2 mL注射器进行尾静脉采血, 血液在4℃冰箱中静止过夜后, 在4℃离心机中离心10min (3500 r/min), 取上清液, 置于–80℃保存待测。随机选取6尾草鱼迅速解剖出肝脏和肠道组织, 其中3尾草鱼的肝脏组织使用多聚甲醛溶液进行固定保存, 另外3尾草鱼的肝脏和肠道组织于1.5 mL无酶离心管中, 置于–80℃保存待测。再随机取3尾草鱼冻存在–20℃冰箱中, 用于体成分的测定。再随机选取4尾草鱼称取鱼体重并解剖再称取内脏重和肝重, 再测量鱼体长, 用于计算肝体比、脏体比和肥满度。
1.4 指标测定及方法
饲料营养成分和体成分测定 水分采用干燥法(GB 5009.3-2016)测定; 粗蛋白用凯氏定氮法(GB 5009.5-2016)测定; 粗脂肪用乙醚索氏提取法(GB 5009.6-2016)测定; 粗灰分用马弗炉(GB 5009.4-2016)测定。游离棉酚含量用苯胺法(GB 13086-91)测定。
生长性能测定 按下列公式计算生长性能指标:
存活率(SR, %)=100×(试验结束鱼尾数/试验开始鱼尾数);
增重率(WGR, %)=100×[(末均重(g)–初均重(g)/初均重(g)];
饲料系数(FCR)=摄食量(g)/[终总重(g)–初总重(g)];
特定生长率(SGR, %/d)=100×[(ln末均重–ln初均重)/养殖天数];
肥满度(CF, g/cm3)=100×终末鱼重(g)/体长(cm)3;
脏体比(VSI)=100×内脏重量(g)/终末鱼重(g);
肝体比(HSI)=100×肝脏重量(g)/终末鱼重(g)。
血清生化指标及免疫指标的测定 血清生化指标低密度脂蛋白胆固醇(LDL-C)、高密度脂蛋白胆固醇(HDL-C)、谷草转氨酶(GOT)、谷丙转氨酶(GPT)、酸性磷酸酶(ACP)、胆汁酸(TBA)、葡萄糖(GLU)、总胆固醇(T-CHO)和甘油三酯(TG)均采用南京建成生物工程研究所的相应试剂盒测定。而补体3 (C3)、补体4 (C4)、免疫球蛋白M(IgM)采用浙江伊利康生物技术有限公司的相应试剂盒测定。
肠道消化酶测定 将肠道从–80℃的冰箱中取出, 准确称取肠道组织进行匀浆后, 测定脂肪酶(Lipase)、淀粉酶(Amylase)及胰蛋白酶(Trypsin)的活性均采用南京建成生物技术有限公司提供的相应试剂盒进行操作。
肝脏组织形态的观察 多聚甲醛溶液固定的肝脏组织经修剪、脱水、包埋、脱蜡、染色、封片后在显微镜下观察切片, 利用CaseViewer 2.2软件截取切片组织照片, 对切片中典型病理改变, 采用箭头标识, 反映出不同组之间的差异。
肝脏抗氧化指标的测定 肝脏组织存于–80℃冰箱中, 在测定前解冻并称重。待测组织用生理盐水进行匀浆, 并在4℃的离心机中离心10min (2500 r/min), 取上清液测定总蛋白含量(TP)及总抗氧化能力(T-AOC)、超氧化物歧化酶(SOD)、丙二醛(MDA)、还原型谷胱甘肽(GSH)和过氧化氢酶(CAT)。以上所有试剂盒均购自南京建成生物工程研究所。
实时荧光定量PCR (qRT-PCR)分析 采用总RNA提取试剂盒(诺唯赞生物科技股份有限公司)提取草鱼肝脏总RNA。采用超微量核酸检测仪对RNA浓度和质量进行检测, RNA样本的A260/A280值在1.8—2.1。使用莫纳生物科技有限公司相关试剂盒将RNA反转录成cDNA。利用Primer Premier 5.0软件进行引物设计, 引物序列见表 2, 引物由上海生工生物股份有限公司合成, 所有引物的扩增效率在0.95—1.10。使用荧光定量PCR仪(LightCycler 48II, Bio-Rad, NUA)和SYBR Premix Ex TaqⅡ试剂盒进行测定。以草鱼β-actin基因作为内参基因, 使用E=2–∆∆Ct计算。
表 2 草鱼抗氧化及免疫相关基因引物Table 2. Primers for antioxidant and immune-related genes in grass carp基因
Gene引物
Primer sequence (5′—3′)登录号
Accession
numberkeap1a GAGCTGATCAGTCAGGACAGTC
GGACTGCAATTGCCTTTGGAGKF811013 nrf2 CCGTTTACCGTGGACATGATC
CACGATGTTCTCCAACTTCCGKX243419 mnsod ATATCAAGAGGCACTGGCCAAG
GAGCCAAAGTCACGCTTAATGGGU218534 gpx1 TGAAAGAGAAGCTGCCTCAACC
GACGATGGTGAGGAACCTTCTGJX854447 gsto CAACGGACAAGATTGGTGCTC
TCTGAGGGTAGACCTCATCCAGKT757314 cat ACGGATCGCACACTTTCAAAC
GTAGAAGGTCCAGGATGGGAAGMG821473 nf-κb AAAAGCAAGCCAATCACAGG
CCGTTTCCTCCTTGAGTTTATCXM051880333.1 tgf-β1 CCGCATAAAGGTCCTACAAAAC
ATGACCCTCACATTCCTACTGCXM051874932 tnf-α ATGATGGAGCATGCCAGTCAG
CTTGTTGAGCGTGAAGCAGACHQ696609 il-8 ATGAGTCTTAGAGGTCTGGGT
ACAGTGAGGGCTAGGAGGGJN663841 il-12β ACAAAGATGAAAAACTGGAGGC
GTGTGTGGTTTAGGTAGGAGCCKF944668.1 il-15 CCTTCCAACAATCTCGCTTC
AACACATCTTCCAGTTCTCCTTKT445872 β-actin GGCTGTGCTGTCCCTGTA
GGGCATAACCCTCGTAGATM25013 1.5 数据统计与分析
本研究的试验数据经过Excel 2019整理后, 使用R (version 4.0.3)包进行数据统计分析。在图和表中, 采用microchat包进行统计学分析。对符合正态性的数据, 进行Shapiro-Wilk正态性检验和Bartlett方差齐性检验, 并采用Student’s t检验进行分析。在结果中, P<0.05表示其他组与CON组之间存在显著差异。另外, P<0.05表示S1组与SQ2组之间存在显著性差异。对于不符合正态性和方差齐性的数据, 采用Wilcoxon秩和检验进行分析。最终的数据呈现为平均值±标准误(mean±SE)。
2. 结果
2.1 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼生长性能的影响
由表 3可知, 与CON组相比, S1和SQ2组草鱼的FBW、WGR和SGR显著降低(P<0.05); 而S1组草鱼的FCR显著升高(P<0.05)。与S1组相比, SQ2组草鱼的FBW、WGR和SGR显著升高(P<0.05), 而FCR显著降低(P<0.05); 其他参数无显著差异(P>0.05)。各处理间草鱼的IBM、SR、CF、HSI和VSI无显著差异(P>0.05)。
表 3 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼生长性能的影响Table 3. Effects of dietary oleanolic acid on the growth performance of grass carp fed high cottonseed and rapeseed meal diet项目Item 组别Group CON S1 SQ2 初均重IBM (g) 50.03±0.09 49.92±0.05 50.11±0.14 末均重FBW (g) 198.07±3.34 180.32±1.67* 188.94±1.69# 增重率WGR (%) 302.23±1.28 259.3±5.30* 277.04±2.57# 特定生长率SGR (%/d) 2.45±0.03 2.29±0.02* 2.37±0.01# 饵料系数FCR 1.21±0.02 1.38±0.05* 1.28±0.01# 存活率SR (%) 99.17±0.83 100.00±0.00 100.00±0.00 肥满度CF (g/cm3) 1.96±0.02 1.90±0.01 1.87±0.02 肝体比HSI 2.42±0.05 2.43±0.11 2.44±0.07 脏体比VSI 16.33±0.41 16.33±0.28 15.34±0.58 注: *P<0.05, 表示其余组与对照组比较有显著性差异, #P<0.05, 表示S1组与SQ2组比较有显著性差异; 下同Note: *P<0.05, indicates a significant difference between the remaining groups and the control group, #P<0.05, indicates a significant difference between the S1 group and SQ2 group; the same applies below 2.2 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼体成分的影响
由表 4可知, 各处理间草鱼的水分、粗蛋白、粗脂肪和粗灰分均无显著差异(P>0.05)。
表 4 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼体成分的影响 (%)Table 4. Effects of dietary oleanolic acid on nutrient composition of grass carp fed high cottonseed and rapeseed meal diet (%)项目Item 组别Group CON S1 SQ2 水分Moisture 73.85±0.19 73.71±0.23 74.04±0.98 粗蛋白Crude protein 14.78±0.43 14.14±0.31 14.80±0.50 粗脂肪Crude lipid 8.94±0.03 8.89±0.05 8.97±0.06 粗灰分Crude ash 2.94±0.11 3.28±0.06 3.20±0.25 2.3 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清生化指标的影响
由表 5可知, 与CON组相比, S1和SQ2组草鱼的血清GOT、TBA和T-CHO的水平显著升高(P<0.05); S1组草鱼的血清LDL-C的含量显著升高(P<0.05)。与S1组相比, SQ2组草鱼的血清GOT和T-CHO的水平显著降低(P<0.05), 而其他参数无显著差异(P>0.05)。各处理间草鱼血清GLU、GPT、TG和HDL-C的水平均无显著差异(P>0.05)。
表 5 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清生化指标的影响Table 5. Effects of dietary oleanolic acid on serum biochemical index of grass carp fed high cottonseed and rapeseed meal diet项目Item 组别Group CON S1 SQ2 葡萄糖GLU (mmol/L) 3.32±0.28 4.13±0.26 3.80±0.03 胆汁酸TBA (µmol/L) 2.88±0.27 4.33±0.06* 3.59±0.05*# 谷草转氨酶GOT (U/L) 17.04±0.96 19.23±0.22* 18.08±0.21*# 谷丙转氨酶GPT (U/L) 49.29±0.30 50.94±0.17 50.26±0.51 总胆固醇T-CHO (mmol/L) 7.03±0.28 7.63±0.06* 6.01±0.20*# 甘油三酯TG (mmol/L) 4.69±0.06 4.67±0.15 4.61±0.11 低密度脂蛋白胆固醇
LDL-C (mmol/L)10.25±0.08 11.53±0.14* 11.06±0.58 高密度脂蛋白胆固醇
HDL-C (mmol/L)11.29±0.28 11.06±0.10 11.07±0.22 2.4 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肠道消化酶的影响
由表 6可知, 与CON组相比, S1和SQ2组草鱼的肠道淀粉酶活性显著降低(P<0.05); S1组草鱼的肠道脂肪酶活性显著降低(P<0.05)。与S1组相比, SQ2组草鱼的肠道淀粉酶和脂肪酶活性显著增高(P<0.05), 并且脂肪酶活性与CON组没有显著差异(P>0.05)。各处理间草鱼的肠道胰蛋白酶活性无显著差异(P>0.05)。
表 6 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肠道消化酶的影响Table 6. Effect of oleanolic acid on intestinal digestive enzymes of grass carp fed high cottonseed and rapeseed meal diet项目 Item 组别Group CON S1 SQ2 胰蛋白酶×103
Trypain (U/mg prot)2.91±0.10 2.44±0.26 2.68±0.03 脂肪酶
Lipase (U/g prot)1.76±0.11 1.42±0.03* 1.73±0.02# 淀粉酶
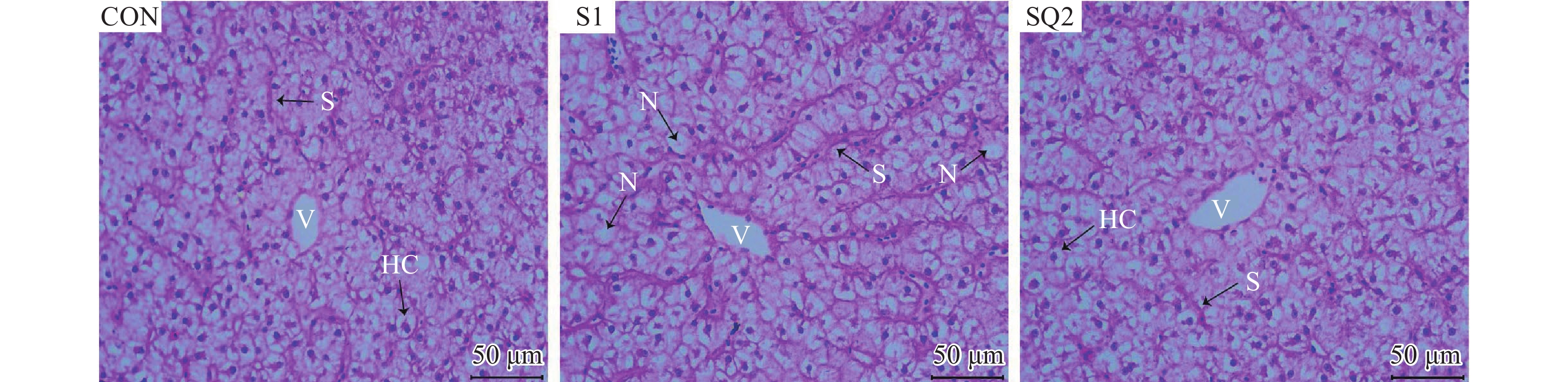
Amylase (U/mg prot)0.65±0.02 0.48±0.01* 0.56±0.03*# 2.5 齐墩果酸对摄食高比例的棉粕和菜粕饲料对草鱼肝脏组织结构的影响
由图 1可知, 通过观察草鱼肝脏组织形态结构发现, CON组草鱼肝细胞核位于近中心位置, 形态轮廓清晰; S1组草鱼肝细胞空泡化, 肝细胞核明显移位; 在高比例的棉粕和菜粕饲料中添加齐墩果酸时, SQ2组草鱼肝细胞的空泡化表现出缓解作用。
2.6 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清免疫指标的影响
由表 7可知, 与CON组相比, S1和SQ2组草鱼的血清ACP和IgM的水平显著降低(P<0.05); S1组草鱼的血清C3的水平显著降低(P<0.05)。与S1组相比, SQ2组草鱼的血清ACP和IgM的水平显著升高(P<0.05), 而C3的水平无显著差异(P>0.05)。各处理间草鱼的血清C4的水平无显著差异(P>0.05)。
表 7 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清免疫指标的影响Table 7. Effects of dietary oleanolic acid on serum immunity of grass carp fed high cottonseed and rapeseed meal diet项目Item 组别Group CON S1 SQ2 酸性磷酸酶ACP
(金氏单位/g prot)21.52±0.31 15.49±0.41* 20.05±0.55# 免疫球蛋白M
IgM (g/L)2.58±0.19 1.05±0.03* 1.69±0.05*# 补体4 C4 (g/L) 0.13±0.02 0.13±0.01 0.14±0.01 补体3 C3 (g/L) 0.34±0.01 0.24±0.00* 0.34±0.01 2.7 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化的影响
由表 8可知, 与CON组相比, S1和SQ2组草鱼的肝脏CAT、SOD、GSH的水平显著降低(P<0.05), 而MDA的含量显著升高(P<0.05); S1组草鱼的肝脏T-AOC的水平显著降低(P<0.05)。与S1组相比, SQ2组草鱼的肝脏的CAT、SOD和T-AOC的水平显著升高(P<0.05), 而MDA的含量显著降低(P<0.05), 其GSH的水平无显著差异(P>0.05)。
表 8 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化的影响Table 8. Effects of dietary oleanolic acid on Antioxidant index of hepatic of grass carp fed high cottonseed and rapeseed meal diet项目Item 组别Group CON S1 SQ2 过氧化氢酶
CAT (U/mg prot)8.83±0.08 6.62±0.14* 7.16±0.05*# 超氧化物歧化酶
SOD (U/mg prot)167.58±0.25 149.96±0.60* 163.32±0.61*# 还原型谷胱甘肽
GSH (U/mg prot)195.43±1.13 163.59±2.68* 170.67±3.06* 总抗氧化能力
T-AOC (U/g prot)63.06±0.81 52.98±1.22* 64.00±1.86# 丙二醛
MDA (nmol/mg prot)3.30±0.16 4.53±0.02* 3.79±0.06*# 2.8 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化相关基因表达的影响
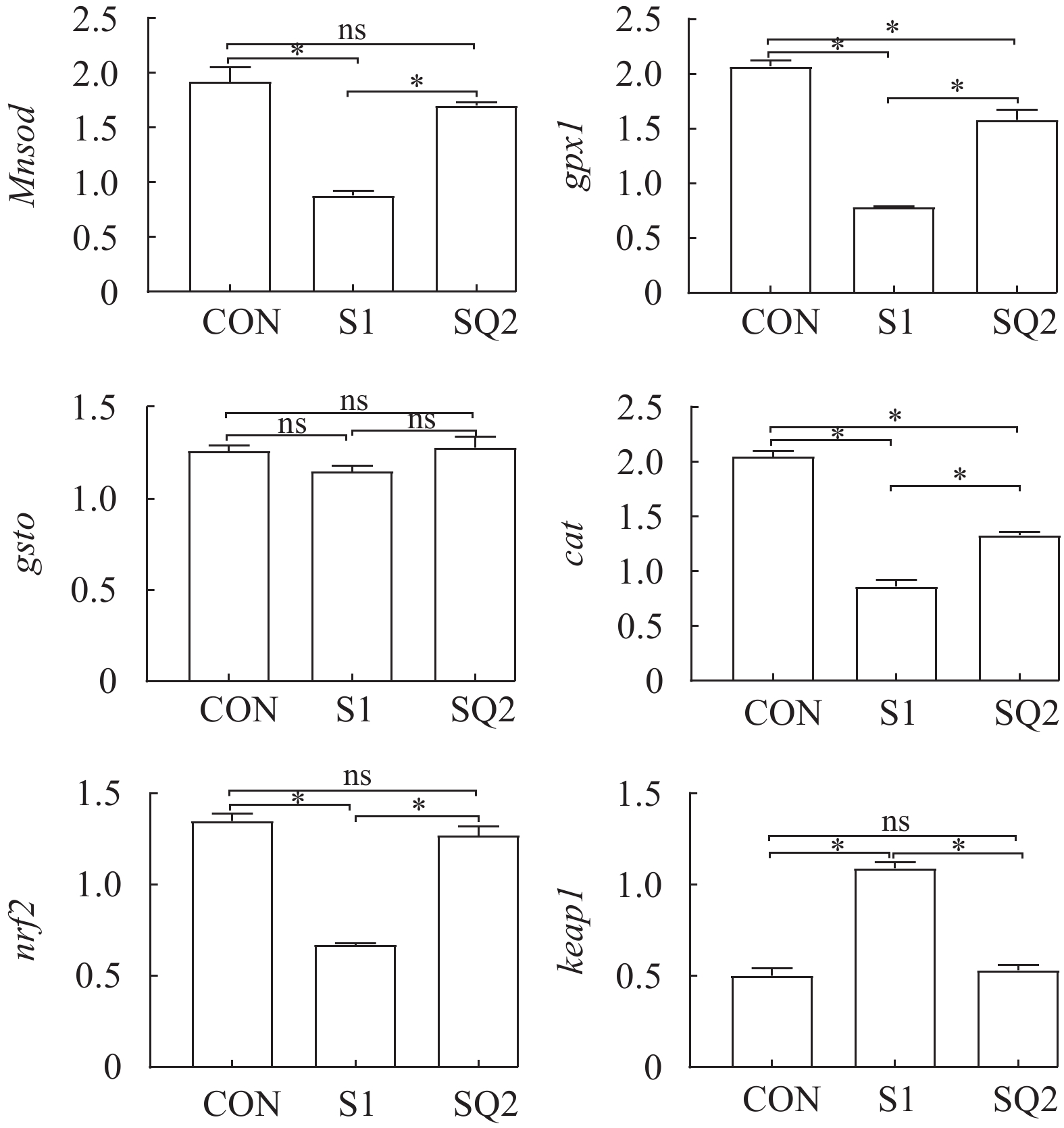
由图 2可知, 与CON组相比, S1组和SQ2组草鱼肝脏中gpx1和cat的表达水平显著下调(P<0.05); S1组草鱼肝脏中Mnsod和nrf2的表达水平显著下调(P<0.05), 而keap1的表达水平显著上调(P<0.05)。与S1组相比, SQ2组草鱼肝脏中Mnsod、gpx1、cat、nrf2的表达水平显著上调(P<0.05), 而keap1的表达水平显著下调(P<0.05)。各处理间草鱼肝脏中gsto的表达水平无显著差异(P>0.05)。
![]() 图 2 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化相关基因表达的影响NS. 表示组间比较无显著差异; *P<0.05表示组间比较有显著差异; 下同Figure 2. Effects of Oleanolic acid to high cottonseed and rapeseed meal diet on liver antioxidant-related gene expression of grass carpNS means that there is no significant difference among groups. *P<0.05 indicates a significant difference among groups; The same applies below
图 2 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化相关基因表达的影响NS. 表示组间比较无显著差异; *P<0.05表示组间比较有显著差异; 下同Figure 2. Effects of Oleanolic acid to high cottonseed and rapeseed meal diet on liver antioxidant-related gene expression of grass carpNS means that there is no significant difference among groups. *P<0.05 indicates a significant difference among groups; The same applies below2.9 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏免疫相关基因表达的影响
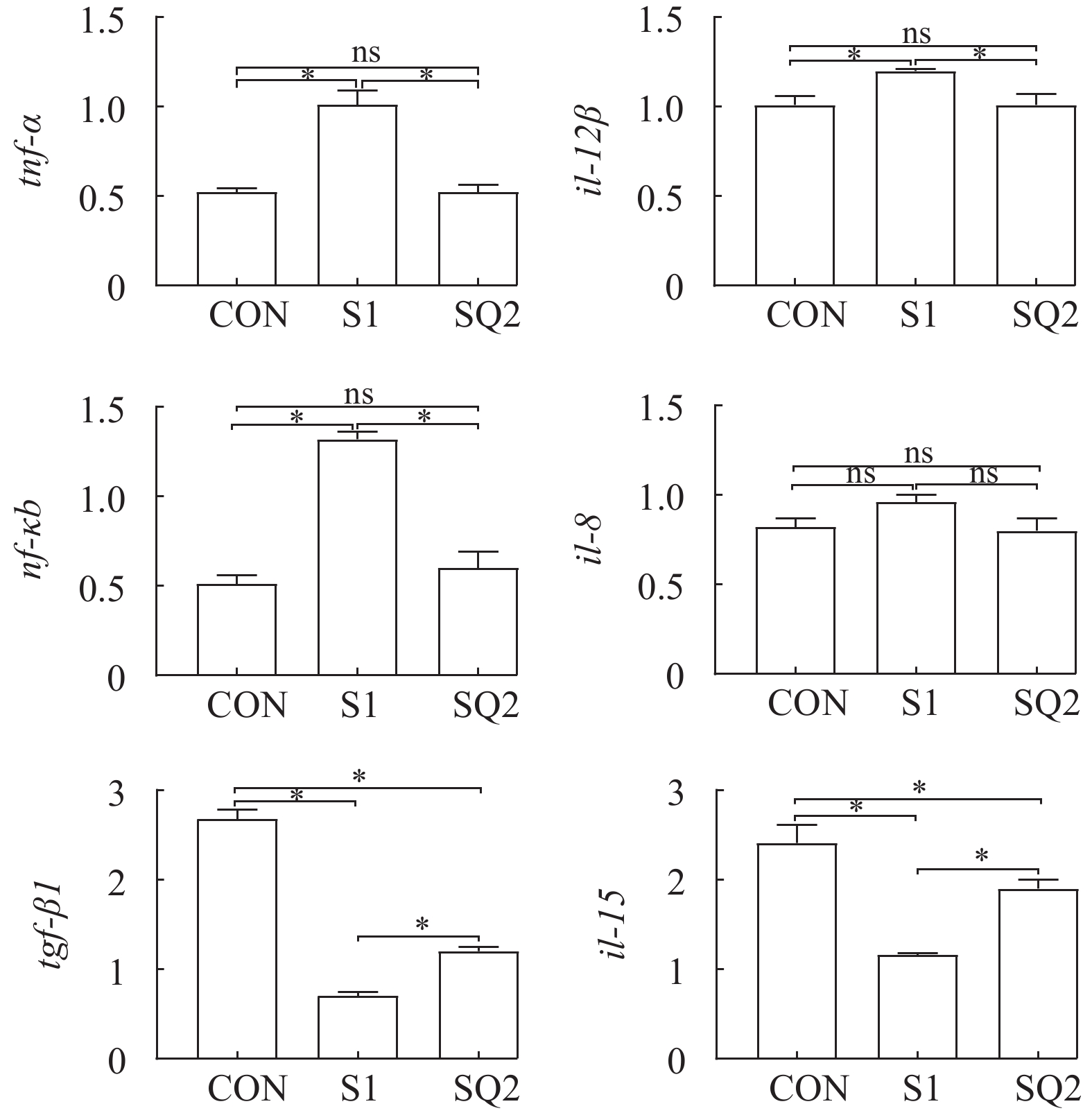
由图 3可知, 与CON组相比, S1组和SQ2组草鱼肝脏中tgf-β1和il-15的表达水平显著下调(P<0.05); S1组草鱼肝脏中tnf-α、nf-κb和il-12β的表达水平显著上调(P<0.05)。与S1组相比, SQ2组草鱼肝脏中tnf-α、nf-κb和il-12β的表达水平显著下调(P<0.05), 而tgf-β1和il-15的表达水平显著上调(P<0.05)。各处理间草鱼肝脏中il-8的表达水平无显著差异(P>0.05)。
3. 讨论
3.1 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼生长性能和体成分的影响
齐墩果酸可促进摄食高比例的棉粕和菜粕的草鱼生长。有研究表明, 饲料中高比例添加棉粕和菜粕会显著影响水产动物的生长性能[12]。本实验结果显示, 长期摄食高棉粕饲料显著降低草鱼的生长性能, 并且提高饲料系数, 但对草鱼的存活率没有显著影响。该结果与草鱼(Ctenopharyngodon idella)[12]、罗非鱼(Oreochromis niloticus)[20]和红鲷(Pagrus major)[10]的研究结果相似, 这可能与棉粕和菜籽粕中的抗氧营养因子有关, 如棉酚、单宁、芥子碱和硫代葡萄糖苷等物质。这些抗营养因子对水产动物的生长性能有显著负面影响[21, 22]。目前, 为了提高植物蛋白源在饲料中的利用率, 有众多研究采用在饲料中添加功能性添加剂的方法, 其中齐墩果酸作为一种天然的植物提取物, 具有消炎抑菌、无抗药性和低毒的特性, 并有研究表明可以促进水产动物的生长性能。本实验结果显示, 在高比例的棉粕和菜粕饲料中添加齐墩果酸能够改善由高比例的棉粕和菜粕饲料造成的草鱼生长性能下降问题, 并且降低饲料系数。该结果与克氏原螯虾(Procambarus clarkia)[19]的研究结果相似。这可能是因为植物提取物富含多种活性物质, 可以增加消化酶活性, 加强草鱼对营养物质的吸收, 从而促进生长[23]。此外, 还有可能是植物提取物可以通过缓解由高棉粕饲料引起的机体损伤(如氧化应激、炎症反应等), 从而促进草鱼的生长[19]。生长可以由机体营养成分侧面反映出来, 但通常鱼体营养成分是处于稳定范围内的。本实验结果表明, 各处理组的草鱼鱼体水分、粗灰分、粗脂肪和粗蛋白含量无显著差异, 但齐墩果酸组草鱼鱼体的粗蛋白有上升的趋势。可能是机体要把更多的能量和物质(合成免疫球蛋白等)用来应对损伤。粗蛋白含量增加, 意味着免疫机能可能得到提升[24]。
3.2 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清生化指标的影响
血清生理生化是可以反映出自身营养代谢状况的重要指标[25]。在血清中, TBA、GOT和GPT是反映肝脏代谢功能障碍的重要指标。GOT和GPT是反映肝脏是否损伤的重要代谢酶, 当肝脏细胞受损时会导致血液转氨酶活性升高[26]。而血清中 TG、TC 是反映机体脂质代谢水平的重要指标, LDL-C 和HDL-C 是反映肝脏脂肪转运情况的重要指标, 当血清含有较高水平的LDL-C能够抑制胆固醇的转运, 不利于机体健康[19]。本实验结果显示, 长期摄食高比例的棉粕和菜粕饲料显著增加草鱼血清中 TBA、GOT、T-CHO 和 LDL-C的水平, 与鲤(Cyprinus carpio)[27]的研究结果相似, 说明棉粕中的棉酚可能对草鱼的肝脏造成了损伤。此外, 在本研究中, 将齐墩果酸添加到高比例的棉粕和菜粕饲料中会显著降低草鱼血清中TBA、GOT和T-CHO的水平。这说明齐墩果酸的添加能够改善草鱼肝脏功能的作用, 该结果与克氏原螯虾[19]和大鼠[28]的研究结果相似。这可能是因为齐墩果酸有利于分解胆汁酸的淤积, 同时增强肝脏代谢功能来对肝脏进行保护作用[28]。
3.3 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清免疫指标的影响
水产动物的体液免疫是非特异性免疫功能的重要组成部分[29]。ACP是一种磷酸水解酶, 是反映鱼类免疫力高低的重要指标[30]。而IgM、C3和C4是补体系统的主要组成成分, 具有补体防御、介导免疫、促进炎症反应和消除病原体等作用[31]。本实验结果显示, 长期摄食高比例的棉粕和菜粕饲料显著降低草鱼血清中ACP、IgM和C3的水平。本实验结果与乌苏里拟鲿幼鱼(Pseudobagrus ussuriensis)[32]和草鱼[21]上的结果相似。此外, 在本研究中, 将齐墩果酸添加到高比例的棉粕和菜粕饲料中显著提高草鱼血清中ACP和IgM的水平。这说明齐墩果酸的添加能够增强草鱼的非特异性免疫功能, 这与克氏原螯虾[19]的研究结果相似。可能是因为齐墩果酸或其体内代谢产物可以激活水产动物的非特异性免疫, 从而刺激碱性磷酸酶、C3和IgM的分泌, 保护水产动物免受伤害。
3.4 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏组织结构的影响
肝脏是鱼类最大的腺体器官, 在营养物质代谢、分泌、排泄、解毒及免疫等方面具有重要的作用[33]。本实验结果显示, 长期摄食高比例的棉粕和菜粕饲料会导致草鱼肝细胞空泡化、细胞核偏移、消失以及出现血窦扩张。进一步研究表明, 在高比例的棉粕和菜粕饲料中添加齐墩果酸可以减少肝细胞空泡化区域, 并缓解血窦扩张。肝脏HE切片显示及血清中TBA、T-CHO的含量和GOT的酶活力呈现出一致性的变化趋势, 进一步验证了齐墩果酸的添加可以改善由高比例的棉粕和菜粕饲料引起的肝脏组织损伤。
3.5 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化的影响
动物组织损伤与氧化损伤密切相关[34]。SOD、CAT和GSH是组成机体酶促抗氧化系统的主要抗氧化酶, 可以保护细胞, 并减少机体受自由基的损伤[35]。T-AOC是机体抗氧化效果的一个重要指标, 具有抵抗体内自由基损伤的重要作用。MDA是脂质过氧化的代谢产物, 其含量的多少可以间接反映活性氧自由基的含量以及脂质过氧化的程度[36]。有研究表明, 饲料中棉粕和菜粕含量过高时, 会导致水产动物产生氧化应激, 其中主要原因是棉菜粕中抗营养因子含量较高[21]。本实验结果发现, 长期摄食高比例的棉粕和菜粕饲料显著降低草鱼肝脏中T-AOC、SOD、CAT和GSH酶活性, 并且显著增加草鱼肝脏MDA的含量。此外, 本研究还发现, 在高比例的棉粕和菜粕饲料中添加齐墩果酸可以显著提高草鱼肝脏中T-AOC、SOD和CAT酶活性, 并且显著降低草鱼肝脏MDA的含量。该结果与克氏原螯虾[19]的研究结果相似, 可能是因为齐墩果酸通过抑制线粒体活性氧来改善高血糖的抵抗, 则表现出降血脂和抗炎作用[37]。
有研究表明, Nrf2/Keap1信号通路在水产动物的氧化应激中起着关键作用, 抗氧化酶的活性受该信号通路的调节[38]。keap1基因负调控nrf2的核转位, 从而抑制抗氧化相关基因的表达[39]。本实验结果表明, 与CON相比, 长期摄食高比例的棉粕和菜粕饲料的草鱼会导致肝脏中gpx1、cat、Mnsod和nrf2的表达水平显著降低, 而keap1的表达水平显著升高。该结果与草鱼[21]的结果相似, 说明棉菜粕中的抗营养因子会通过Nrf2/Keap1信号通路, 降低抗氧化酶相关基因表达, 从而引起水产动物抗氧化功能紊乱, 导致氧化损伤。此外, 本研究还发现, 在高比例的棉粕和菜粕饲料中添加齐墩果酸可以显著提高草鱼肝脏中gpx1、cat、Mnsod和nrf2表达水平, 并且显著降低草鱼肝脏keap1的表达水平, 表明Nrf2被激活, 并调控下游基因, 提高机体的抗氧化能力。并有研究发现, 齐墩果酸对小鼠肝脏的保护作用与Nrf2/Keap1可能相关[40]。
3.6 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏免疫相关基因的影响
炎症反应是机体免疫系统应对外界抗原的重要组成部分, 主要由细胞因子调控炎症反应的发生[41]。有研究表明, NF-κB在调节免疫应答的过程起关键作用, 当NF-κB被激活后, 会启动靶基因表达如il-12β、il-8和tnf-α等促炎因子[42]。同时抑制抗炎因子的产生, 如il-15和tgf-β1等, 从而会导致组织结构损伤, 严重危害鱼类机体健康[43]。本实验结果表明, 与CON相比, 长期摄食高比例的棉粕和菜粕饲料的草鱼会导致肝脏中nf-κb、tnf-α和il-12β的表达水平显著升高, 而il-15和tgf-β1的表达水平显著降低。该结果与草鱼[21]的结果相似, 说明高比例的棉粕和菜粕饲料会通过激活NF-κB信号通路, 促进草鱼肝脏促炎因子的分泌, 从而引起炎症反应。进一步研究表明, 在高比例的棉粕和菜粕饲料中添加齐墩果酸可以显著降低草鱼肝脏中nf-κb、tnf-α和il-12β表达水平, 并且显著提高草鱼肝脏il-15和tgf-β1的表达水平。可能是因为齐墩果酸可以通过靶向NF-κB并抑制其活化, 以及促炎因子的表达[44, 45]。
4. 结论
饲料中过高比例的棉菜粕显著降低了草鱼的生长性能、机体抗氧化能力和免疫力, 造成草鱼肝脏氧化损伤和炎症反应。齐墩果酸的添加促进了鱼体蛋白质的沉积, 并缓解草鱼肝脏氧化应激诱发的炎症反应, 提高草鱼的生长性能、抗氧化能力和免疫力。
-
图 2 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化相关基因表达的影响
NS. 表示组间比较无显著差异; *P<0.05表示组间比较有显著差异; 下同
Figure 2. Effects of Oleanolic acid to high cottonseed and rapeseed meal diet on liver antioxidant-related gene expression of grass carp
NS means that there is no significant difference among groups. *P<0.05 indicates a significant difference among groups; The same applies below
表 1 饲料组成和营养水平(干物质基础, %)
Table 1 Composition and nutritional level of diets (DM basis, %)
原料Ingredient 组别Group CON S1 SQ2 鱼粉Fish meal 2.00 0 0 豆粕Soybean meal 27.00 0 0 棉粕Cottonseed meal 12.00 28.00 28.00 菜粕Rapeseed meal 12.00 27.00 27.00 面粉Wheat flour 20.00 20.00 20.00 米糠Rice bran 10.00 10.00 10.00 麦麸Wheat bran 3.76 1.36 1.31 豆油Soybean oil 2.50 2.90 2.90 干酒糟及其可溶物DDGS 8.00 8.00 8.00 胆碱Choline 0.20 0.20 0.20 磷酸二氢钙Ca(H2PO4)2 1.50 1.50 1.50 预混料Premixa 1.00 1.00 1.00 防霉剂Antimold 0.03 0.03 0.03 抗氧化剂Antioxidants 0.01 0.01 0.01 齐墩果酸Oleanolic acid 0 0 0.05 合计Total 100.00 100.00 100.00 营养成分Nutrient levelsb 粗蛋白Crude protein 31.22 31.12 31.14 粗脂肪Crude lipid 6.78 6.64 6.81 粗灰分Crude ash 7.20 7.28 7.27 游离棉酚Free gossypol (mg/kg) 99.81 233.52 231.17 注: a预混料来源于青岛玛斯特生物技术有限公司, 成分参考本课题组前期研究[19]; b营养组成为测定值Note: aPremixes are obtained from Qingdao Mast Bio-technology Co., Ltd. and the ingredients are referred to the previous research of this group[19]; bProximate composition are measured values 表 2 草鱼抗氧化及免疫相关基因引物
Table 2 Primers for antioxidant and immune-related genes in grass carp
基因
Gene引物
Primer sequence (5′—3′)登录号
Accession
numberkeap1a GAGCTGATCAGTCAGGACAGTC
GGACTGCAATTGCCTTTGGAGKF811013 nrf2 CCGTTTACCGTGGACATGATC
CACGATGTTCTCCAACTTCCGKX243419 mnsod ATATCAAGAGGCACTGGCCAAG
GAGCCAAAGTCACGCTTAATGGGU218534 gpx1 TGAAAGAGAAGCTGCCTCAACC
GACGATGGTGAGGAACCTTCTGJX854447 gsto CAACGGACAAGATTGGTGCTC
TCTGAGGGTAGACCTCATCCAGKT757314 cat ACGGATCGCACACTTTCAAAC
GTAGAAGGTCCAGGATGGGAAGMG821473 nf-κb AAAAGCAAGCCAATCACAGG
CCGTTTCCTCCTTGAGTTTATCXM051880333.1 tgf-β1 CCGCATAAAGGTCCTACAAAAC
ATGACCCTCACATTCCTACTGCXM051874932 tnf-α ATGATGGAGCATGCCAGTCAG
CTTGTTGAGCGTGAAGCAGACHQ696609 il-8 ATGAGTCTTAGAGGTCTGGGT
ACAGTGAGGGCTAGGAGGGJN663841 il-12β ACAAAGATGAAAAACTGGAGGC
GTGTGTGGTTTAGGTAGGAGCCKF944668.1 il-15 CCTTCCAACAATCTCGCTTC
AACACATCTTCCAGTTCTCCTTKT445872 β-actin GGCTGTGCTGTCCCTGTA
GGGCATAACCCTCGTAGATM25013 表 3 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼生长性能的影响
Table 3 Effects of dietary oleanolic acid on the growth performance of grass carp fed high cottonseed and rapeseed meal diet
项目Item 组别Group CON S1 SQ2 初均重IBM (g) 50.03±0.09 49.92±0.05 50.11±0.14 末均重FBW (g) 198.07±3.34 180.32±1.67* 188.94±1.69# 增重率WGR (%) 302.23±1.28 259.3±5.30* 277.04±2.57# 特定生长率SGR (%/d) 2.45±0.03 2.29±0.02* 2.37±0.01# 饵料系数FCR 1.21±0.02 1.38±0.05* 1.28±0.01# 存活率SR (%) 99.17±0.83 100.00±0.00 100.00±0.00 肥满度CF (g/cm3) 1.96±0.02 1.90±0.01 1.87±0.02 肝体比HSI 2.42±0.05 2.43±0.11 2.44±0.07 脏体比VSI 16.33±0.41 16.33±0.28 15.34±0.58 注: *P<0.05, 表示其余组与对照组比较有显著性差异, #P<0.05, 表示S1组与SQ2组比较有显著性差异; 下同Note: *P<0.05, indicates a significant difference between the remaining groups and the control group, #P<0.05, indicates a significant difference between the S1 group and SQ2 group; the same applies below 表 4 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼体成分的影响 (%)
Table 4 Effects of dietary oleanolic acid on nutrient composition of grass carp fed high cottonseed and rapeseed meal diet (%)
项目Item 组别Group CON S1 SQ2 水分Moisture 73.85±0.19 73.71±0.23 74.04±0.98 粗蛋白Crude protein 14.78±0.43 14.14±0.31 14.80±0.50 粗脂肪Crude lipid 8.94±0.03 8.89±0.05 8.97±0.06 粗灰分Crude ash 2.94±0.11 3.28±0.06 3.20±0.25 表 5 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清生化指标的影响
Table 5 Effects of dietary oleanolic acid on serum biochemical index of grass carp fed high cottonseed and rapeseed meal diet
项目Item 组别Group CON S1 SQ2 葡萄糖GLU (mmol/L) 3.32±0.28 4.13±0.26 3.80±0.03 胆汁酸TBA (µmol/L) 2.88±0.27 4.33±0.06* 3.59±0.05*# 谷草转氨酶GOT (U/L) 17.04±0.96 19.23±0.22* 18.08±0.21*# 谷丙转氨酶GPT (U/L) 49.29±0.30 50.94±0.17 50.26±0.51 总胆固醇T-CHO (mmol/L) 7.03±0.28 7.63±0.06* 6.01±0.20*# 甘油三酯TG (mmol/L) 4.69±0.06 4.67±0.15 4.61±0.11 低密度脂蛋白胆固醇
LDL-C (mmol/L)10.25±0.08 11.53±0.14* 11.06±0.58 高密度脂蛋白胆固醇
HDL-C (mmol/L)11.29±0.28 11.06±0.10 11.07±0.22 表 6 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肠道消化酶的影响
Table 6 Effect of oleanolic acid on intestinal digestive enzymes of grass carp fed high cottonseed and rapeseed meal diet
项目 Item 组别Group CON S1 SQ2 胰蛋白酶×103
Trypain (U/mg prot)2.91±0.10 2.44±0.26 2.68±0.03 脂肪酶
Lipase (U/g prot)1.76±0.11 1.42±0.03* 1.73±0.02# 淀粉酶
Amylase (U/mg prot)0.65±0.02 0.48±0.01* 0.56±0.03*# 表 7 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼血清免疫指标的影响
Table 7 Effects of dietary oleanolic acid on serum immunity of grass carp fed high cottonseed and rapeseed meal diet
项目Item 组别Group CON S1 SQ2 酸性磷酸酶ACP
(金氏单位/g prot)21.52±0.31 15.49±0.41* 20.05±0.55# 免疫球蛋白M
IgM (g/L)2.58±0.19 1.05±0.03* 1.69±0.05*# 补体4 C4 (g/L) 0.13±0.02 0.13±0.01 0.14±0.01 补体3 C3 (g/L) 0.34±0.01 0.24±0.00* 0.34±0.01 表 8 齐墩果酸对摄食高比例的棉粕和菜粕饲料草鱼肝脏抗氧化的影响
Table 8 Effects of dietary oleanolic acid on Antioxidant index of hepatic of grass carp fed high cottonseed and rapeseed meal diet
项目Item 组别Group CON S1 SQ2 过氧化氢酶
CAT (U/mg prot)8.83±0.08 6.62±0.14* 7.16±0.05*# 超氧化物歧化酶
SOD (U/mg prot)167.58±0.25 149.96±0.60* 163.32±0.61*# 还原型谷胱甘肽
GSH (U/mg prot)195.43±1.13 163.59±2.68* 170.67±3.06* 总抗氧化能力
T-AOC (U/g prot)63.06±0.81 52.98±1.22* 64.00±1.86# 丙二醛
MDA (nmol/mg prot)3.30±0.16 4.53±0.02* 3.79±0.06*# -
[1] Dawood M A O, Abo-Al-Ela H G, Hasan M T. Modulation of transcriptomic profile in aquatic animals: Probiotics, prebiotics and synbiotics scenarios [J]. Fish & Shellfish Immunology, 2020(97): 268-282.
[2] Wang T, Xu M, Wang J, et al. A combination of rapeseed, cottonseed and peanut meal as a substitute of soybean meal in diets of Yellow River carp Cyprinus carpio var [J]. Aquaculture Nutrition, 2020, 26(5): 1520-1532. doi: 10.1111/anu.13099
[3] Wang J, Clark G, Ju M, et al. Effects of replacing menhaden fishmeal with cottonseed flour on growth performance, feed utilization and body composition of juvenile red drum Sciaenops ocellatus [J]. Aquaculture, 2020(523): 735217.
[4] Kang P, Hang Y, Chen C, et al. Effects of replacing fishmeal with rapeseed meal and dietary condensed tannins on antioxidant capacity, immunity, and hepatic and intestinal health of largemouth bass (Micropterus salmoides) [J]. Aquaculture Reports, 2023(30): 101548.
[5] Coutinho F, Simões R, Monge-Ortiz R, et al. Effects of dietary methionine and taurine supplementation to low-fish meal diets on growth performance and oxidative status of European sea bass (Dicentrarchus labrax) juveniles [J]. Aquaculture, 2017(479): 447-454.
[6] Wang K Z, Feng L, Jiang W D, et al. Dietary gossypol reduced intestinal immunity and aggravated inflammation in on-growing grass carp (Ctenopharyngodon idella) [J]. Fish & Shellfish Immunology, 2019(86): 814-831.
[7] Tan Q, Liu Q, Chen X, et al. Growth performance, biochemical indices and hepatopancreatic function of grass carp, Ctenopharyngodon idellus, would be impaired by dietary rapeseed meal [J]. Aquaculture, 2013(414-415): 119-126.
[8] von Danwitz A, Schulz C. Effects of dietary rapeseed glucosinolates, sinapic acid and phytic acid on feed intake, growth performance and fish health in turbot (Psetta maxima L.) [J]. Aquaculture, 2020(516): 734624.
[9] Dossou S, Koshio S, Ishikawa M, et al. Effect of partial replacement of fish meal by fermented rapeseed meal on growth, immune response and oxidative condition of red sea bream juvenile, Pagrus major [J]. Aquaculture, 2018(490): 228-235.
[10] Dossou S, Koshio S, Ishikawa M, et al. Growth performance, blood health, antioxidant status and immune response in red sea bream (Pagrus major) fed Aspergillus oryzae fermented rapeseed meal (RM-Koji) [J]. Fish & Shellfish Immunology, 2018(75): 253-262.
[11] Zhu B P, Zhou J, Wang Z, et al. Interactions between intestinal morphology, digestion, inflammatory responses, and gut microbiota of juvenile channel catfish elicited by dietary enzymatic rice protein [J]. Fish & Shellfish Immunology, 2022(127): 155-165.
[12] Liu Y L, Zhong L, Chen T, et al. Dietary sanguinarine supplementation on the growth performance, immunity and intestinal health of grass carp (Ctenopharyngodon idellus) fed cottonseed and rapeseed meal diets [J]. Aquaculture, 2020(528): 735521.
[13] Pu H, Li X, Du Q, et al. Research progress in the application of Chinese herbal medicines in aquaculture: a review [J]. Engineering, 2017, 3(5): 731-737. doi: 10.1016/J.ENG.2017.03.017
[14] Peng X, Li X, Li Y, et al. The protective effect of oleanolic acid on NMDA-induced MLE-12 cells apoptosis and lung injury in mice by activating SIRT1 and reducing NF-κB acetylation [J]. International Immunopharmacology, 2019(70): 520-529.
[15] Chibber P, Kumar C, Singh A, et al. Anti-inflammatory and analgesic potential of OA-DHZ; a novel semisynthetic derivative of dehydrozingerone [J]. International Immunopharmacology, 2020(83): 106469.
[16] Xin C, Liu S, Qu H, et al. The novel nano complexes containing deoxycholic acid-grafted chitosan and oleanolic acid displays the hepatoprotective effect against CCl(4)-induced liver injury in vivo [J]. International Journal of Biological Macromolecules, 2021(185): 338-349.
[17] Jia W, Liang S, Jin M, et al. Oleanolic acid inhibits hypoxic tumor-derived exosomes-induced premetastatic niche formation in hepatocellular carcinoma by targeting ERK1/2-NFκB signaling [J]. Phytomedicine, 2024(126): 155208.
[18] Wang Y P, Lu Y, Zhang Y, et al. The draft genome of the grass carp (Ctenopharyngodon idellus) provides insights into its evolution and vegetarian adaptation [J]. Nature Genetics, 2015, 47(6): 625-631. doi: 10.1038/ng.3280
[19] Fu G, Wang J, Cai M, et al. Beneficial effects of oleanolic acid on hepatopancreas oxidative stress and intestinal microbiota structure of red swamp crayfish (Procambarus clarkia) [J]. Aquaculture Reports, 2023(33): 101797.
[20] Wu F, Tian J, Yu L, et al. Effects of dietary rapeseed meal levels on growth performance, biochemical indices and flesh quality of juvenile genetically improved farmed tilapia [J]. Aquaculture Reports, 2021(20): 100679.
[21] Shi Y, Liu Y, Xie K, et al. Dietary sanguinarine ameliorates growth impairment and intestinal dysfunction in Ctenopharyngodon idellus fed a high cottonseed meal diet [J]. Aquaculture, 2023(576): 739864.
[22] Collins S A, Øverland M, Skrede A, et al. Effect of plant protein sources on growth rate in salmonids: Meta-analysis of dietary inclusion of soybean, pea and canola/rapeseed meals and protein concentrates [J]. Aquaculture, 2013(400/401): 85-100.
[23] Xie J J, Chen X, Guo T Y, et al. Dietary values of Forsythia suspensa extract in Penaeus monodon under normal rearing and Vibrio parahaemolyticus 3HP (VP(3HP)) challenge conditions: effect on growth, intestinal barrier function, immune response and immune related gene expression [J]. Fish & Shellfish Immunology, 2018(75): 316-326.
[24] Liu Y, Hu Z J, Xing H Y, et al. Renoprotective effects of oleanolic acid and its possible mechanisms in rats with diabetic kidney disease [J]. Biochemical and Biophysical Research Communications, 2022(636): 1-9.
[25] Ahmed I, Reshi Q M, Fazio F. The influence of the endogenous and exogenous factors on hematological parameters in different fish species: a review [J]. Aquaculture International, 2020, 28(3): 869-899. doi: 10.1007/s10499-019-00501-3
[26] Li J, Xu W, Lai W, et al. Effect of dietary methionine on growth performance, lipid metabolism and antioxidant capacity of large yellow croaker (Larimichthys crocea) fed with high lipid diets [J]. Aquaculture, 2021(536): 736388.
[27] Wang X F, Li X Q, Leng X J, et al. Effects of dietary cottonseed meal level on the growth, hematological indices, liver and gonad histology of juvenile common carp (Cyprinus carpio) [J]. Aquaculture, 2014(428/429): 79-87.
[28] Liu J, Liu J, Meng C, et al. Oleanolic acid alleviates ANIT-induced cholestatic liver injury by activating Fxr and Nrf2 pathways to ameliorate disordered bile acids homeostasis [J]. Phytomedicine, 2022(102): 154173.
[29] Ni P J, Jiang W D, Wu P, et al. Dietary low or excess levels of lipids reduced growth performance, and impaired immune function and structure of head kidney, spleen and skin in young grass carp (Ctenopharyngodon idella) under the infection of Aeromonas hydrophila [J]. Fish & Shellfish Immunology, 2016(55): 28-47.
[30] Wang J T, Han T, Li X Y, et al. Effects of dietary phosphatidylcholine (PC) levels on the growth, molt performance and fatty acid composition of juvenile swimming crab, Portunus trituberculatus [J]. Animal Feed Science and Technology, 2016(216): 225-233.
[31] Ichiki S, Kato-Unoki Y, Somamoto T, et al. The binding spectra of carp C3 isotypes against natural targets independent of the binding specificity of their thioester [J]. Developmental and Comparative Immunology, 2012, 38(1): 10-16. doi: 10.1016/j.dci.2012.03.004
[32] Bu X, Chen A, Lian X, et al. An evaluation of replacing fish meal with cottonseed meal in the diet of juvenile Ussuri catfish Pseudobagrus ussuriensis: growth, antioxidant capacity, nonspecific immunity and resistance to Aeromonas hydrophila [J]. Aquaculture, 2017(479): 829-837.
[33] Cao J, Chen J, Wang J, et al. Effects of fluoride on liver apoptosis and Bcl-2, Bax protein expression in freshwater teleost, Cyprinus carpio [J]. Chemosphere, 2013, 91(8): 1203-1212. doi: 10.1016/j.chemosphere.2013.01.037
[34] Yu H H, Qiu Y X, Li B, et al. Kadsura heteroclita stem ethanol extract protects against carbon tetrachloride-induced liver injury in mice via suppression of oxidative stress, inflammation, and apoptosis [J]. Journal of Ethnopharmacology, 2021(267): 113496.
[35] 张姚铮泰, 谢凯, 石勇, 等. 饲料蛋白质和脂肪水平对丝尾鳠幼鱼生长、血清生化指标及肝脏抗氧化能力的影响 [J]. 水生生物学报, 2024(1): 1-12. doi: 10.7541/2023.2023.0093 Zhang Y Z T, Xie K, Shi Y, et al. Dietary protein and lipid level on drowth, serum biochemical index and liver antioxidant capacity of juvenil asian red-tailed catfish (Hemibagrus wychioides) [J]. Acta Hydrobiologica Sinica, 2024(1): 1-12. doi: 10.7541/2023.2023.0093
[36] Ren Y, Men X, Yu Y, et al. Effects of transportation stress on antioxidation, immunity capacity and hypoxia tolerance of rainbow trout (Oncorhynchus mykiss) [J]. Aquaculture Reports, 2022(22): 100940.
[37] Wang X, Liu R, Zhang W, et al. Oleanolic acid improves hepatic insulin resistance via antioxidant, hypolipidemic and anti-inflammatory effects [J]. Molecular and Cellular Endocrinology, 2013, 376(1/2): 70-80.
[38] Shi Y, Zhong L, Liu Y L, et al. Effects of dietary andrographolide levels on growth performance, antioxidant capacity, intestinal immune function and microbioma of rice field eel (Monopterus Albus) [J]. Animals, 2020, 10(10).
[39] Jung K A, Kwak M K. The Nrf2 system as a potential target for the development of indirect antioxidants [J]. Molecules, 2010, 15(10): 7266-7291. doi: 10.3390/molecules15107266
[40] Lu Y F, Liu J, Wu K C, et al. Protection against phalloidin-induced liver injury by oleanolic acid involves Nrf2 activation and suppression of Oatp1b2 [J]. Toxicology Letters, 2015, 232(1): 326-332. doi: 10.1016/j.toxlet.2014.09.027
[41] Chen S, Zeng J, Li R, et al. Traditional Chinese medicine in regulating macrophage polarization in immune response of inflammatory diseases [J]. Journal of Ethnopharmacology, 2024(325): 117838.
[42] Yang M, Wang Y, Patel G, et al. In vitro and in vivo anti-inflammatory effects of different extracts from Epigynum auritum through down-regulation of NF-κB and MAPK signaling pathways [J]. Journal of Ethnopharmacology, 2020(261): 113105.
[43] Shi Y, Zhong L, Fan Y, et al. The protective effect of mulberry leaf flavonoids on high-carbohydrate-induced liver oxidative stress, inflammatory response and intestinal microbiota disturbance in Monopterus albus [J]. Antioxidants, 2022, 11(5): 976. doi: 10.3390/antiox11050976
[44] Hwang Y J, Song J, Kim H R, et al. Oleanolic acid regulates NF-κB signaling by suppressing MafK expression in RAW 264.7 cells [J]. BMB Reports, 2014, 47(9): 524-529. doi: 10.5483/BMBRep.2014.47.9.149
[45] Yang Y H, Dai S Y, Deng F H, et al. Recent advances in medicinal chemistry of oleanolic acid derivatives [J]. Phytochemistry, 2022(203): 113397.



 下载:
下载: