THE INFLUENCE OF STOCKING DENSITY ON THE GROWTH, BLOOD BIOCHEMISTRY, AND ANTIOXIDANT CAPACITY INDICATORS OF CHINESE PERCH (SINIPERCA CHUATSI)
-
摘要: 研究旨在探讨不同养殖密度对翘嘴鳜(Siniperca chuatsi)生长、血液生化和抗氧化能力指标的影响。在循环水系统的水箱中(60 cm×60 cm×40 cm)于5种密度下[超低养殖密度组(ELD; 3.34 kg/m3)、低养殖密度组(LD; 9.51 kg/m3)、中等养殖密度组(MD; 15.82 kg/m3)、高养殖密度组(HD; 21.05 kg/m3)和超高养殖密度组(EHD; 25.99 kg/m3)]养殖翘嘴鳜[平均体重(175.76±15.85) g]40d。结果表明, ELD组和LD组在增重率(WGR)和饲料转化率(FCR)方面表现出更好的生产性能(P<0.05), 并增加血浆超氧化物歧化酶(SOD)、过氧化氢酶(CAT)和谷胱甘肽过氧化物酶(GSH-Px)的活性, 同时减少丙二醛(MDA)的形成(P<0.05)。此外, EHD组的血浆葡萄糖水平、总胆固醇水平和甘油三酯水平均较高(P<0.05)。LD组刺鼠基因相关蛋白(agrp)的mRNA表达下降, 而ELD组阿片促黑色素皮质激素原(pomc)基因表达下调。综上, 推荐在循环系统中养殖翘嘴鳜的适宜放养密度为3.34—9.51 kg/m3。Abstract: The present study was to explore the influence of stocking density on the growth, blood biochemistry, and antioxidant capacity indicators of Chinese perch (Siniperca chuatsi) cultured in the bank (60 cm× 60 cm×40 cm) based recirculating systems for 40d. Fish (average weight 175.76±15.85 g) were reared in triplicate under five densities: extra-low stocking density (ELD; 3.34 kg/m3); low stocking density (LD; 9.51 kg/m3); medium stocking density (MD; 15.82 kg/m3); high stocking density (HD; 21.05 kg/m3) and extra-high stocking density (EHD; 25.99 kg/m3). After 40 days, growth performance, biochemical parameters, appetite-related genes expression and stress indicators of fish were evaluated. The results showed that the ELD and LD groups showed better productive performance for weight gain (WG) and feed conversion ratio (FCR) (P<0.05), and increased plasma superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GSH-Px) activities, and decreased the formation of malondialdehyde (MDA) (P<0.05). Additionally, the EHD group performed higher glucose levels, cholesterol levels and lower triglyceride levels in plasma (P<0.05). The mRNA expression of agouti gene-related protein (agrp) was riesd in LD group, whereas gene expressions of pro-opiomelanocortin (pomc) resulted in a down-regulation in ELD group. In sum, the suitable stocking density for culturing Chinese perch in recirculating systems is recommended to be between 3.34 and 9.51 kg/m3. It is of great significance to select the best stocking density for improving the economic benefit of fish culture.
-
Keywords:
- Stocking density /
- Growth /
- Appetite /
- Stress /
- Siniperca chuatsi
-
渔业和水产养殖在提供粮食、营养和就业方面发挥着越来越重要的作用[1]。中国是世界上最大的水产养殖生产国和出口国, 其水产养殖业已成为粮食安全的主要参与者之一[1, 2]。随着中国经济水平的提高, 为满足日益增长的水产品需求, 水产养殖从粗放式向集约式转变[3]。在集约化养殖中, 诸多因素影响着鱼类的发育、健康和福利, 而养殖密度往往被认为是影响单位面积养殖产量和经济效益的重要因素[4-6]。
生长与组织的化学、生化和功能变化密切相关, 是评价水产养殖效率的关键参数之一。Shao等[7]认为, 循环水养殖系统下高饲养密度(HSD)养殖的石斑鱼具有更好的聚集和摄食行为。在大西洋鲑(Salmo salar L.)和非洲鲶(Claras gariepinus)中也发现了类似的结果[8, 9]。相比之下, 在高放养水平下饲养的尼罗罗非鱼(Oreochromis niloticus)生长缓慢[10]。持续的HSD养殖可能会减缓鱼的生长, 增加对疾病的敏感性, 并引起应激反应[11]。HSD饲养条件下的异育银鲫(Carassius gibelio)生长受到明显抑制, 而低养殖密度(LSD)组的生长速度最高[12]。然而, 由于空间利用率低, LSD可能导致生产成本增加和盈利能力下降[7]。此外, 在鱼类中不适当的放养密度是导致应激反应的原因之一, 它可能会干扰一些物种的代谢途径[13, 14]。高饲养密度所引起的应激会促使血浆生化参数的慢性升高, 限制鱼类的生长, 最终影响水产养殖的盈利能力[15]。因此, 选择最佳养殖密度对提高养殖经济效益具有重要意义。
翘嘴鳜(Sinperca chuatsi)是我国优良的商业水产养殖品种, 口感鲜美, 经济价值高[16, 17]。在鱼苗开始进食后, 拒绝吃死饵料或混合饵料, 只接受活饵料[16, 18]。本研究初步探讨了不同养殖密度对循环系统中翘嘴鳜生长、食欲及应激的影响, 以期筛选出适宜的养殖密度, 为今后的养殖生产提供参考。
1. 材料与方法
1.1 实验鱼和实验设计
实验鱼(175.76±15.85) g来自鳜鱼研究中心(武汉, 中国), 试验在华中农业大学水产学院(武汉, 中国)进行。根据我们之前的研究, 在养殖过程中实验鱼经驯化后摄食人工饲料[19]。随后将实验鱼转入循环系统中[温度: (22±0.5)℃, 溶解氧: (9±0.5) mg/L]适应实验条件2周。随后根据5个养殖密度对每个养殖缸的鱼数量进行了调整: 超低密度组(ELD; 3.34 kg/m3; 4条鱼/缸)、低密度组(ELD; 9.51 kg/m3; 10 条鱼/缸)、中密度组(MD; 15.82 kg/m3; 16 条鱼/缸)和高密度组(HD; 21.05 kg/m3); 超高密度组(EHD; 25.99 kg/m3; 28 条鱼/缸), 每组3个重复。所有试验鱼均按5%体重的试验饲料饲喂, 每日2次(09:00和17:00), 每次反复投喂3遍, 投喂至桶底出现残饵且鱼不再摄食为止, 投喂完毕后收集残饵, 烘干称重, 计算每天摄食饲料量(FI)。试验周期为40d, 实验前后统计每个养殖缸鱼的数量。试验饲料的主要原料购自武汉高龙饲料科技有限公司, 并在实验室制粒机(CPM)中通过模具进行精细研磨、充分混合和制粒。试验饲料的主要营养成分如表 1所示。
表 1 不同水平碳水化合物水平饲料组成成分Table 1. Compositions of diets added with different levels of carbohydrate原料种类Ingredient (g 100/g diet) 含量Content 鱼粉Fish meal1 70.0 玉米淀粉 Corn starch2 20.0 鱼油Fish oil 3.0 矿物质预混料Mineral premix3 2.0 维生素预混料Vitamin premix4 2.0 羧甲基纤维素钠Carboxymethylcellulose sodium 3.0 总计Total 100 基本成分Proximate composition 干物质Dry matter (% DM) 84.4 粗蛋白Crude protein 47.2 粗脂肪Crude lipid 6.7 碳水化合物Carbohydrate 27.5 灰分Ash 18.6 总能Gross energy (kJ/g) 13.6 注: 1. 鱼粉的粗蛋白质和碳水化合物含量分别为67%和7%; 2. 玉米淀粉的粗蛋白质和粗脂肪含量分别为0.3%和0.2%; 3. 矿物质预混料(每千克饲料): 硫酸锰, 10 mg; 硫酸镁, 10 mg; 氯化钾, 95 mg; 氯化钠, 165 mg; 硫酸锌, 20 mg; 氯化碘, 1 mg; 硫酸铜, 12.5 mg; 硫酸亚铁, 105 mg; 亚硒酸钠, 0.1 mg; 钴, 1.5 mg; 4. 维生素预混料(每千克饲料): 维生素A, 2000国际单位; 维生素B1 (硫胺素), 5 mg; 维生素B2(核黄素), 5 mg; 维生素B6, 5 mg; 维生素B12, 0.025 mg; 维生素D3, 1200国际单位; 维生素E, 21 mg; 维生素K3, 2.5 mg; 叶酸, 1.3 mg; 生物素, 0.05 mg; 泛酸钙, 20 mg; 肌醇, 60 mg; 抗坏血酸(35%), 110 mg; 烟酰胺, 25 mgNote: 1. Crude protein and carbohydrate content of fish meal are 67% and 7%, respectively; 2. Crude protein and crude lipid content of corn starch are 0.3% and 0.2%, respectively; 3. Mineral premix (per kg of diet): MnSO4, 10 mg; MgSO4, 10 mg; KCl, 95 mg; NaCl, 165 mg; ZnSO4, 20 mg; KI, 1 mg; CuSO4,12.5 mg; FeSO4, 105 mg; Na2SeO3, 0.1 mg; Co, 1.5 mg; 4. Vitamin premix (per kg of diet): vitamin A, 2000 IU; vitamin B1 (thiamin), 5 mg; vitamin B2 (riboflavin), 5 mg; vitamin B6, 5 mg; vitamin B12, 0.025 mg; vitamin D3, 1200 IU; vitamin E 21 mg; vitamin K3 2.5 mg; folic acid, 1.3 mg; biotin, 0.05 mg; pantothenic acid calcium, 20 mg; inositol, 60 mg; ascorbic acid. (35%), 110 mg; niacinamide, 25 mg 摄食量(FI, g/d)=饲料摄入量/实验天数。
1.2 样品采集
养殖结束后, 实验鱼禁食24h取样, 在取样前用150 mg/L MS-222(Sigma, 美国)麻醉。每个鱼缸的所有实验鱼都称量它们的最终重量来计算增重率(WG)和饵料系数(FCR)。每个鱼缸随机抽取2尾鱼进行静脉血采样(每组6尾), 立即取血样于4℃ 3000×g离心15min, 采集血浆进行分析。下丘脑用液氮冷冻, –80℃保存, 用于定量检测。
增重率(WGR, %)=100×(最终重量–初始重量)/初始重量;
日采食量(FI, g/d)=饲料摄入量/实验天数;
饵料系数(FCR)=饲料摄入量/鱼体增重;
存活率(SR, %)=100×实验开始鱼尾数/实验结束鱼尾数。
1.3 血液生化指标的测定
血浆中葡萄糖、总甘油三酯和总胆固醇的水平使用南京建城生物技术研究所生产葡萄糖测定试剂盒(Catalog No. F006-1-1)、总甘油三酯检测试剂盒(Catalog No. A110-1-1)和总胆固醇测定试剂盒(Catalog No. A111-1-1), 并遵循生产商的操作指南。
1.4 定量分析
按照RNAiso从每个样品中提取总RNA, 用HiScript II 1st Strand cDNA Synthesis Kit (Vazyme, 中国)反转录合成第一链cDNA。每个反应体系(20 μL)包含1 μL cDNA模板、10 μL SYBR(Vazyme, 中国)、引物各0.4和8.2 μL ddH2O。在本研究中使用的两个食欲基因序列见表 2。反应条件: 95℃10s, 95℃5s, 58℃30s, 40个循环, 溶解曲线(从65℃以0.5℃/s速率逐渐升到95℃, 每隔6s采集一次数据信号)。rpl13a基因作为内参基因, 使用2–ΔΔCt值法测定基因的相对表达量[20]。
表 2 用于定量实时PCR的引物序列Table 2. Primer sequences for the quantitative real-time PCR引物Primer 序列
Sequence (5′—3′)退火温度
Tm (℃)序列号
Accession numbersagrp F GTGCTGCTCTGCTGTTGG 58 NM_001328012.1 agrp R AGGTGTCACAGGGGTCGC pomc F GGCTGAAGATGGTGTGTCTATG 58 NM_001083051.1 pomc R ACATGCAGAGGTGAATACAGTC rpl13a F CACCCTATGACAAGAGGAAGC 58 MK770673 rpl13a R TGTGCCAGACGCCCAAG 1.5 应激参数
本研究测量了超氧化物歧化酶(SOD)、过氧化氢酶(CAT)、谷胱甘肽过氧化物酶(GSH-Px)和丙二醛(MDA)四个应激参数。血浆中超氧化物歧化酶(SOD)活性测定试剂盒(Catalog No. A001-1)、过氧化氢酶(CAT)活性测定试剂盒(Catalog No. A007-1)、谷胱甘肽过氧化物酶(GSH-Px)活性测定试剂盒(Catalog No. A005-1)和丙二醛(MDA)含量测定试剂盒(Catalog No. A003-1)均购自南京建城生物技术研究所, 并遵循生产商指南。
1.6 数据分析
所有数据均表示为均值标准差(Mean standard error of the mean, SD), 数据的正态分布首先通过Shapiro-Wilk检验, 组间均数比较采用单因素方差分析(ANOVA)和Tukey检验。显著性水平设为P<0.05。
2. 结果
2.1 生长性能
如表 3所示, 增重率随放养密度的增加呈下降趋势。ELD组的增重最高(P<0.05), 饲料系数最低(P<0.05), 与此同时, MD组的食物摄入量最高(P<0.05)。
表 3 翘嘴鳜的生长性能Table 3. Growth parameters of Chinese perch项目Item 增重率WGR (%) 摄食量FI (g/d) 饵料系数FCR 存活率SR (%) ELD
(3.34 kg/m3)52.10±1.50b 115.63±5.21a 1.34±0.08a 100 LD
(9.51 kg/m3)49.00±1.12b 109.50±4.75a 1.40±0.05a 100 MD
(15.82 kg/m3)18.80±1.15a 148.59±4.46a 4.04±0.03b 100 HD
(21.05 kg/m3)15.70±1.48a 125.91±2.71a 4.24±0.09b 100 EHD
(25.99 kg/m3)14.85±2.00a 120.00±3.98a 4.40±0.05b 100 2.2 血液生化指标
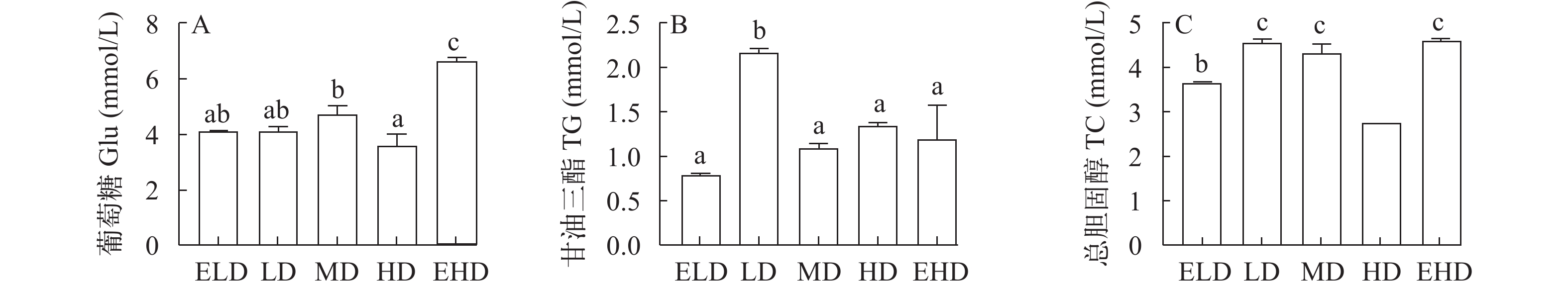
如图 1所示, EHD组的血液葡萄糖水平最高(P<0.05)(图 1A), 最显著的甘油三酯水平出现在LD组(P<0.05; 图 1B), 对于总胆固醇水平, LD、MD和EHD组的值较高(P<0.05; 图 1C)。
![]() 图 1 不同饲养密度对血浆中葡萄糖(A)、甘油三酯(B)和总胆固醇(C)水平的影响值用平均数±标准差(n=6)来表示, 不同上标字母表示实验组间差异显著(P<0.05); 下同Figure 1. Effects of different stocking densities on glucose (A), triglycerides (B) and cholesterol (C) in the plasmaData are represented as mean±SD with 6 replicates, different superscript letters show significant differences between the experimental groups (P<0.05). The same applies below
图 1 不同饲养密度对血浆中葡萄糖(A)、甘油三酯(B)和总胆固醇(C)水平的影响值用平均数±标准差(n=6)来表示, 不同上标字母表示实验组间差异显著(P<0.05); 下同Figure 1. Effects of different stocking densities on glucose (A), triglycerides (B) and cholesterol (C) in the plasmaData are represented as mean±SD with 6 replicates, different superscript letters show significant differences between the experimental groups (P<0.05). The same applies below2.3 食欲相关基因表达
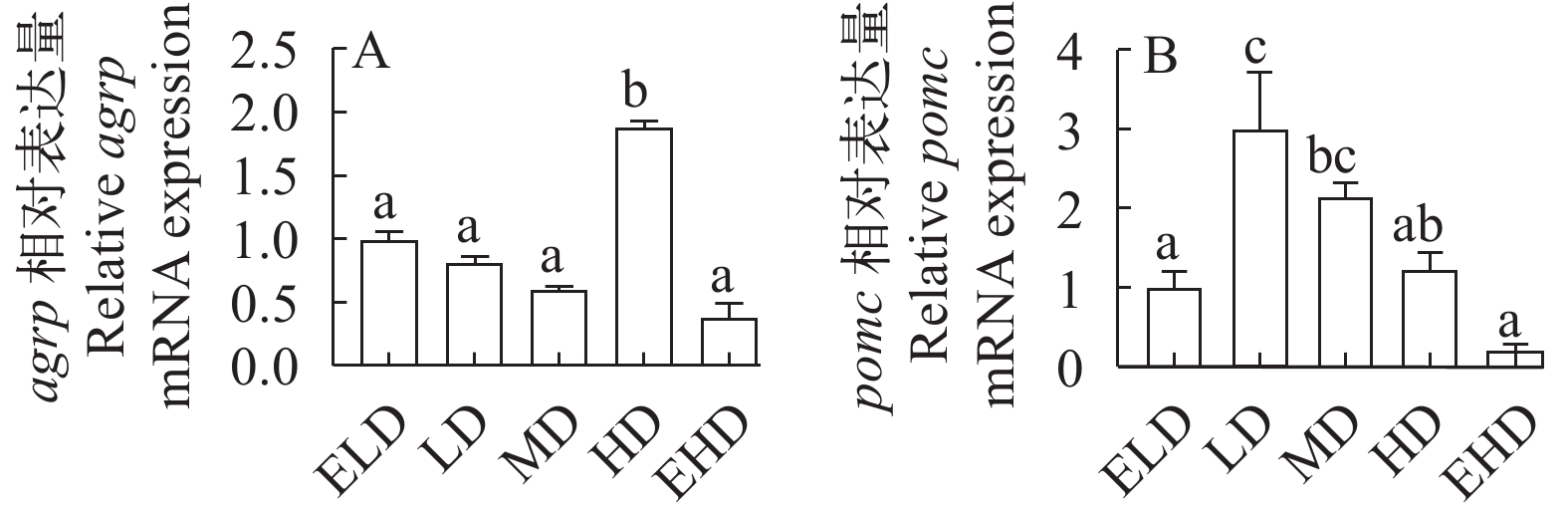
如图 2所示, HD组的刺鼠基因相关蛋白(agrp)mRNA表达显著高于ELD、LD、MD和EHD组(P<0.05; 图 2A)。LD组下丘脑阿黑皮素原基因(pomc)表达显著高于ELD和EHD组(P<0.05; 图 2B)。
2.4 应激指标
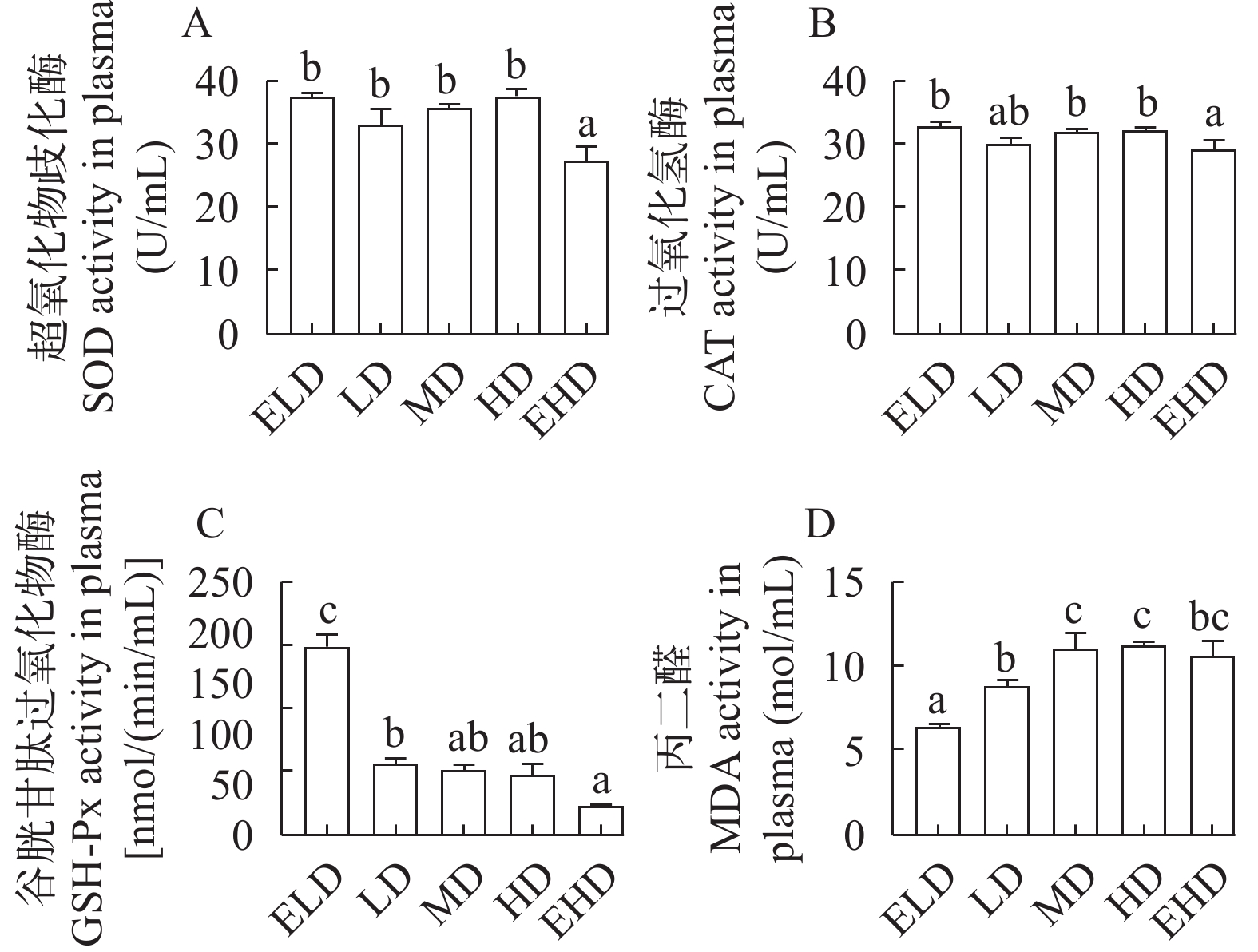
如图 3所示, EHD组血浆SOD和CAT活性显著低于其他各组(P<0.05)。相反, 随着密度的增加, GSH-Px活性呈下降趋势(图 3C)。MDA活性随饲养密度的增加而显著升高, HD组MDA活性最高(P<0.05), EHD组MDA活性较低(图 3D)。
3. 讨论
在本研究中, 养殖密度显著影响鳜鱼的生长速度、食欲基因表达水平和抗氧化能力, 并改变了葡萄糖、甘油三酯和总胆固醇水平。影响鱼类生长性能的主要因素是摄食和营养吸收[21]。事实上, 更高的密度也许更能提高鱼类的生长性能, 因为鱼群的凝聚力、行为和摄食反应更好[7, 22]。有报道称在低养殖密度分别为1.47、4.80和12.18 kg/m3的异育银鲫(Carassius gibelio)、中华鲟(Acipenser sinensis)和斑点叉尾鮰(Ictalurus punctatus)的生长得到了促进[12, 23, 24]。然而, 在本研究中, 翘嘴鳜在ELD组(3.34 kg/m3)和LD组(9.51 kg/m3)显示较高的增重率(WG)和更好的饲料转化率(FCR; 表 3)。随着养殖密度的增加, 翘嘴鳜的WG和FCR逐渐降低, 且高密度组、中密度组和低密度组间差异显著, 说明高密度养殖对鳜鱼的发育有不利影响。这可能是由于空间的增加和低密度养殖鱼对饲料的竞争减少, 同时高密度养殖也会引起应激反应进而影响鳜鱼的食欲。
血浆葡萄糖水平可作为鱼类的应激指标[25]。在本研究中, EHD养殖的鱼获得了较高的血液葡萄糖值水平, 这可能与拥挤的应激有关。EHD组葡萄糖和总胆固醇水平升高可能与糖原分解途径的糖异生和能量代谢有关[15]。大菱鲆(Scophthalmus maximus)、金头鲷(Sparus auratus)、牙鲆(Paralichthys olivaceus)、麦瑞加拉鲮(Cirrhinus mrigala)和塞内加尔鳎(Solea senegalensis)血浆中的葡萄糖水平升高, 表明在高饲养密度紧张的条件下, 有效能量增加[26—30]。此外, 甘油三酯是一种能量来源, 以适应不适当的放养密度。在本研究中, 养殖密度高组的甘油三酯水平均高于ELD组[31, 32]。
在硬骨鱼中, 相关研究表明, 在其中心和外围存在脂肪酸和葡萄糖传感器系统, 可通过食欲和促食欲神经肽调节食物摄入[33, 34]。刺鼠基因相关蛋白(AgRP)是一种可调节食物摄入的食欲生成肽[35], 而促厌食性的阿片促黑色素皮质激素原(POMC)是下丘脑食欲控制的关键因素[36]。在本研究中, HD组显著提高了agrp的表达水平。此外, 我们还分析了pomc的mRNA表达随着密度水平的增加呈先显著上调后下调的趋势, 表明不同的养殖密度可能会影响鳜鱼的食欲。
当抗氧化防御系统无法中和活性氧时, 过量的活性氧会引起氧化应激[37]。生物体通过酶促抗氧化防御保护系统(如SOD、CAT、GSH-Px和MDA)来避免或修复组织损伤, 这些系统被用作氧化应激的指标[38]。与ELD组比较, EHD组SOD、CAT和GSH-Px降低。虹鳟和银鲫体内SOD、CAT和GSH-Px的含量也随着养殖密度的增加而降低[39]。此外, 脂质氧化的终产物(MDA)随着密度的增加而增加, 表明在3.34 kg/m3的ELD环境下, 翘嘴鳜能完全消除外源自由基。
综上, 本研究揭示了养殖密度对翘嘴鳜生长、血液生化特征、食欲和应激的影响。高密度组导致鱼类应激并抑制了其生长。在高放养密度养殖下, 升高的葡萄糖和总胆固醇水平可能会抑制翘嘴鳜的生长。同时不同养殖密度对食物摄入的作用可能与agrp和pomc有关。因此, 推荐在水循环系统中翘嘴鳜的适宜放养密度在3.34—9.51 kg/m3。
-
图 1 不同饲养密度对血浆中葡萄糖(A)、甘油三酯(B)和总胆固醇(C)水平的影响
值用平均数±标准差(n=6)来表示, 不同上标字母表示实验组间差异显著(P<0.05); 下同
Figure 1. Effects of different stocking densities on glucose (A), triglycerides (B) and cholesterol (C) in the plasma
Data are represented as mean±SD with 6 replicates, different superscript letters show significant differences between the experimental groups (P<0.05). The same applies below
表 1 不同水平碳水化合物水平饲料组成成分
Table 1 Compositions of diets added with different levels of carbohydrate
原料种类Ingredient (g 100/g diet) 含量Content 鱼粉Fish meal1 70.0 玉米淀粉 Corn starch2 20.0 鱼油Fish oil 3.0 矿物质预混料Mineral premix3 2.0 维生素预混料Vitamin premix4 2.0 羧甲基纤维素钠Carboxymethylcellulose sodium 3.0 总计Total 100 基本成分Proximate composition 干物质Dry matter (% DM) 84.4 粗蛋白Crude protein 47.2 粗脂肪Crude lipid 6.7 碳水化合物Carbohydrate 27.5 灰分Ash 18.6 总能Gross energy (kJ/g) 13.6 注: 1. 鱼粉的粗蛋白质和碳水化合物含量分别为67%和7%; 2. 玉米淀粉的粗蛋白质和粗脂肪含量分别为0.3%和0.2%; 3. 矿物质预混料(每千克饲料): 硫酸锰, 10 mg; 硫酸镁, 10 mg; 氯化钾, 95 mg; 氯化钠, 165 mg; 硫酸锌, 20 mg; 氯化碘, 1 mg; 硫酸铜, 12.5 mg; 硫酸亚铁, 105 mg; 亚硒酸钠, 0.1 mg; 钴, 1.5 mg; 4. 维生素预混料(每千克饲料): 维生素A, 2000国际单位; 维生素B1 (硫胺素), 5 mg; 维生素B2(核黄素), 5 mg; 维生素B6, 5 mg; 维生素B12, 0.025 mg; 维生素D3, 1200国际单位; 维生素E, 21 mg; 维生素K3, 2.5 mg; 叶酸, 1.3 mg; 生物素, 0.05 mg; 泛酸钙, 20 mg; 肌醇, 60 mg; 抗坏血酸(35%), 110 mg; 烟酰胺, 25 mgNote: 1. Crude protein and carbohydrate content of fish meal are 67% and 7%, respectively; 2. Crude protein and crude lipid content of corn starch are 0.3% and 0.2%, respectively; 3. Mineral premix (per kg of diet): MnSO4, 10 mg; MgSO4, 10 mg; KCl, 95 mg; NaCl, 165 mg; ZnSO4, 20 mg; KI, 1 mg; CuSO4,12.5 mg; FeSO4, 105 mg; Na2SeO3, 0.1 mg; Co, 1.5 mg; 4. Vitamin premix (per kg of diet): vitamin A, 2000 IU; vitamin B1 (thiamin), 5 mg; vitamin B2 (riboflavin), 5 mg; vitamin B6, 5 mg; vitamin B12, 0.025 mg; vitamin D3, 1200 IU; vitamin E 21 mg; vitamin K3 2.5 mg; folic acid, 1.3 mg; biotin, 0.05 mg; pantothenic acid calcium, 20 mg; inositol, 60 mg; ascorbic acid. (35%), 110 mg; niacinamide, 25 mg 表 2 用于定量实时PCR的引物序列
Table 2 Primer sequences for the quantitative real-time PCR
引物Primer 序列
Sequence (5′—3′)退火温度
Tm (℃)序列号
Accession numbersagrp F GTGCTGCTCTGCTGTTGG 58 NM_001328012.1 agrp R AGGTGTCACAGGGGTCGC pomc F GGCTGAAGATGGTGTGTCTATG 58 NM_001083051.1 pomc R ACATGCAGAGGTGAATACAGTC rpl13a F CACCCTATGACAAGAGGAAGC 58 MK770673 rpl13a R TGTGCCAGACGCCCAAG 表 3 翘嘴鳜的生长性能
Table 3 Growth parameters of Chinese perch
项目Item 增重率WGR (%) 摄食量FI (g/d) 饵料系数FCR 存活率SR (%) ELD
(3.34 kg/m3)52.10±1.50b 115.63±5.21a 1.34±0.08a 100 LD
(9.51 kg/m3)49.00±1.12b 109.50±4.75a 1.40±0.05a 100 MD
(15.82 kg/m3)18.80±1.15a 148.59±4.46a 4.04±0.03b 100 HD
(21.05 kg/m3)15.70±1.48a 125.91±2.71a 4.24±0.09b 100 EHD
(25.99 kg/m3)14.85±2.00a 120.00±3.98a 4.40±0.05b 100 -
[1] FAO. The state of world fisheries and aquaculture. Sustainability in action. Rome. 2020. https://doi.org/10.4060/ca9229en.
[2] Mo W Y, Chen Z, Leung H M, et al. Application of veterinary antibiotics in China’s aquaculture industry and their potential human health risks [J]. Environmental Science and Pollution Research International, 2017, 24(10): 8978-8989. doi: 10.1007/s11356-015-5607-z
[3] Yogev U, Barnes A, Giladi I, et al. Potential environmental impact resulting from biased fish sampling in intensive aquaculture operations [J]. Science of the Total Environment, 2020(707): 135630. doi: 10.1016/j.scitotenv.2019.135630
[4] Castillo-Vargasmachuca S, Ponce-Palafox J T, García-Ulloa M, et al. Effect of stocking density on growth performance and yield of subadult pacific red snapper cultured in floating sea cages [J]. North American Journal of Aquaculture, 2012, 74(3): 413-418. doi: 10.1080/15222055.2012.676002
[5] Ellis T, North B, Scott A P, et al. The relationships between stocking density and welfare in farmed rainbow trout [J]. Journal of Fish Biology, 2002, 61(3): 493-531. doi: 10.1111/j.1095-8649.2002.tb00893.x
[6] Ni M, Wen H, Li J, et al. The physiological performance and immune responses of juvenile Amur sturgeon (Acipenser schrenckii) to stocking density and hypoxia stress [J]. Fish & Shellfish Immunology, 2014, 36(2): 325-335. doi: 10.1016/j.fsi.2013.12.002
[7] Shao T, Chen X, Zhai D, et al. Evaluation of the effects of different stocking densities on growth and stress responses of juvenile hybrid grouper ♀ Epinephelus fuscoguttatus × ♂ Epinephelus lanceolatus in recirculating aquaculture systems [J]. Journal of Fish Biology, 2019, 95(4): 1022-1029. doi: 10.1111/jfb.14093
[8] Fenderson O C, Carpenter M R. Effects of crowding on the behaviour of juvenile hatchery and wild landlocked Atlantic salmon (Salmo salar L.) [J]. Animal Behaviour, 1971, 19(3): 439-447. doi: 10.1016/S0003-3472(71)80096-9
[9] van de Nieuwegiessen P G, Ramli N M, Knegtel B P F J M, et al. Coping strategies in farmed African catfish Clarias gariepinus. Does it affect their welfare [J]? Journal of Fish Biology, 2010, 76(10): 2486-2501. doi: 10.1111/j.1095-8649.2010.02635.x
[10] Zaki M A A, Alabssawy A N, Nour A E A M, et al. The impact of stocking density and dietary carbon sources on the growth, oxidative status and stress markers of Nile tilapia (Oreochromis niloticus) reared under biofloc conditions [J]. Aquaculture Reports, 2020, 16(4): 100282. doi: 10.1016/j.aqrep.2020.100282
[11] Sadhu N, Sharma S R K, Joseph S, et al. Chronic stress due to high stocking density in open sea cage farming induces variation in biochemical and immunological functions in Asian seabass (Lates calcarifer, Bloch) [J]. Fish Physiology and Biochemistry, 2014, 40(4): 1105-1113. doi: 10.1007/s10695-014-9909-8
[12] Onxayvieng K, Piria M, Fuka M M, et al. High stocking density alters growth performance, blood biochemical profiles, and hepatic antioxidative capacity in gibel carp (Carassius gibelio) [J]. Fish Physiology and Biochemistry, 2021, 47(2): 203-212. doi: 10.1007/s10695-020-00905-6
[13] Braun N, de Lima R L, Baldisserotto B, et al. Growth, biochemical and physiological responses of Salminus brasiliensis with different stocking densities and handling [J]. Aquaculture, 2010, 301(1/2/3/4): 22-30. doi: 10.1016/j.aquaculture.2010.01.022
[14] Ren Y, Wen H, Li Y, et al. Effects of stocking density on lipid deposition and expression of lipid-related genes in Amur sturgeon (Acipenser schrenckii) [J]. Fish Physiology and Biochemistry, 2017, 43(6): 1707-1720. doi: 10.1007/s10695-017-0403-y
[15] Barton B A, Iwama G K. Physiological changes in fish from stress in aquaculture with emphasis on the response and effects of corticosteroids [J]. Annual Review of Fish Diseases, 1991, 1(1): 3-26. doi: 10.1016/0959-8030(91)90019-G
[16] Liang X F, Oku H, Ogata H Y, et al. Weaning Chinese perch Siniperca chuatsi (Basilewsky) onto artificial diets based upon its specific sensory modality in feeding [J]. Aquaculture Research, 2008(32): 76-82. doi: 10.1046/j.1355-557x.2001.00006.x
[17] Liu H Z, Chen Y Y. Phylogeny of the sinipercine fishes with some taxonomic notes [J]. Zoological Research, 1994, 15(S1): 1-12.
[18] 梁旭方. 鳜鱼视觉特性及其对捕食习性适应的研究Ⅱ. 视网膜结构特性 [J]. 水生生物学报, 1994, 18(4): 376-377. doi: 10.3321/j.issn:1000-3207.1994.04.009 Liang X F. Visual characteristics of mandarin fish (Siniperca chuatsi) in relation to its feeding habit: ii. general properties of the Retina [J]. Acta Hydrobiologica Sinica, 1994, 18(4): 376-377. doi: 10.3321/j.issn:1000-3207.1994.04.009
[19] Zhang Y, Zhang Z, Liang X F, et al. Response of g6p homologous genes in Chinese perch to high-carbohydrate diets [J]. Aquaculture Reports, 2021(19): 100581. doi: 10.1016/j.aqrep.2020.100581
[20] Livak K J, Schmittgen T D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method [J]. Methods, 2001, 25(4): 402-408. doi: 10.1006/meth.2001.1262
[21] Kitagawa A T, Costa L S, Paulino R R, et al. Feeding behavior and the effect of photoperiod on the performance and hematological parameters of the pacamã catfish (Lophiosilurus alexandri) [J]. Applied Animal Behaviour Science, 2015(171): 211-218. doi: 10.1016/j.applanim.2015.08.025
[22] Hayat M, Nugroho R A, Aryani R. Influence of different stocking density on the growth, feed efficiency, and survival of Majalaya common carp (Cyprinus carpio Linnaeus 1758) [J]. F1000Research, 2018(7): 1917.
[23] Long L, Zhang H, Ni Q, et al. Effects of stocking density on growth, stress, and immune responses of juvenile Chinese sturgeon (Acipenser sinensis) in a recirculating aquaculture system [J]. Comparative Biochemistry and Physiology. Toxicology & Pharmacology, 2019(219): 25-34. doi: 10.1016/j.cbpc.2019.02.002
[24] Refaey M M, Li D, Tian X, et al. High stocking density alters growth performance, blood biochemistry, intestinal histology, and muscle quality of channel catfish Ictalurus punctatus [J]. Aquaculture, 2018(492): 73-81. doi: 10.1016/j.aquaculture.2018.04.003
[25] Martinez-Porchas M, Martinez-Cordova L R, Ramos-Enriquez R. Cortisol and glucose: reliable indicators of fish stress [J]? Pan-American Journal of Aquatic Sciences, 2009, 4(2): 158-178.
[26] Bolasina S, Tagawa M, Yamashita Y, et al. Effect of stocking density on growth, digestive enzyme activity and cortisol level in larvae and juveniles of Japanese flounder, Paralichthys olivaceus [J]. Aquaculture, 2006, 259(1/2/3/4): 432-443. doi: 10.1016/j.aquaculture.2006.05.021
[27] Liu B, Jia R, Han C, et al. Effects of stocking density on antioxidant status, metabolism and immune response in juvenile turbot (Scophthalmus maximus) [J]. Comparative Biochemistry and Physiology. Part C : Toxicology & Pharmacology, 2016(190): 1-8. doi: 10.1016/j.cbpc.2016.07.007
[28] Mancera J M, Vargas-Chacoff L, García-López A, et al. High density and food deprivation affect arginine vasotocin, isotocin and melatonin in gilthead sea bream (Sparus auratus) [J]. Comparative Biochemistry and Physiology. Part A : Molecular & Integrative Physiology, 2008, 149(1): 92-97. doi: 10.1016/j.cbpa.2007.10.016
[29] Tejpal C S, Pal A K, Sahu N P, et al. Dietary supplementation of l-tryptophan mitigates crowding stress and augments the growth in Cirrhinus mrigala fingerlings [J]. Aquaculture, 2009, 293(3/4): 272-277. doi: 10.1016/j.aquaculture.2008.09.014
[30] Andrade T, Afonso A, Pérez-Jiménez A, et al. Evaluation of different stocking densities in a Senegalese sole (Solea senegalensis) farm: implications for growth, humoral immune parameters and oxidative status [J]. Aquaculture, 2015(438): 6-11. doi: 10.1016/j.aquaculture.2014.12.034
[31] Herrera M, Vargas-Chacoff L, Hachero I, et al. Physiological responses of juvenile wedge sole Dicologoglossa cuneata (Moreau) to high stocking density [J]. Aquaculture Research, 2009, 40(7): 790-797. doi: 10.1111/j.1365-2109.2008.02162.x
[32] Laiz-Carrión R, Viana I R, Cejas J R, et al. Influence of food deprivation and high stocking density on energetic metabolism and stress response in red porgy, Pagrus pagrus L [J]. Aquaculture International, 2012, 20(3): 585-599. doi: 10.1007/s10499-011-9488-y
[33] Conde-Sieira M, Agulleiro M J, AguilarA J, et al. Effect of different glycaemic conditions on gene expression of neuropeptides involved in control of food intake in rainbow trout; interaction with stress [J]. Journal of Experimental Biology, 2010, 213(22): 3858-3865. doi: 10.1242/jeb.048439
[34] Polakof S, Mommsen T P, Soengas J L. Glucosensing and glucose homeostasis: from fish to mammals [J]. Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 2011, 160(4): 123-149. doi: 10.1016/j.cbpb.2011.07.006
[35] Engström Ruud L, Pereira M M A, de Solis A J, et al. NPY mediates the rapid feeding and glucose metabolism regulatory functions of AgRP neurons [J]. Nature Communications, 2020, 11(1): 442. doi: 10.1038/s41467-020-14291-3
[36] Morton G J, Cummings D E, Baskin D G, et al. Central nervous system control of food intake and body weight [J]. Nature, 2006, 443(7109): 289-295.
[37] Kohen R, Nyska A. Oxidation of biological systems: oxidative stress phenomena, antioxidants, redox reactions, and methods for their quantification [J]. Toxicologic Pathology, 2002, 30(6): 620-650. doi: 10.1080/01926230290166724
[38] Halliwell B, Gutteridge J M C. Free radicals in biology and medicine [J]. Journal of Free Radicals in Biology & Medicine, 1985, 1(4): 331-332. doi: 10.1016/0748-5514(85)90140-0
[39] Sahin K, Yazlak H, Orhan C, et al. The effect of lycopene on antioxidant status in rainbow trout (Oncorhynchus mykiss) reared under high stocking density [J]. Aquaculture, 2014(418/419): 132-138. doi: 10.1016/j.aquaculture.2013.10.009
-
期刊类型引用(5)
1. 张现红,李文洋,刘宝良,费凡,高小强,郭冉,曹淑全,朱智文. 密度胁迫对珍珠龙胆石斑鱼生长和生理的影响. 渔业科学进展. 2025(01): 71-81 .  百度学术
百度学术
2. 王大伟,邢盈,蒋成宇,胡淼,赵金良. 放养密度对鳜幼鱼HPI轴激素、应激酶活力及呼吸频率的影响. 淡水渔业. 2025(02): 26-32 .  百度学术
百度学术
3. 林秀蔚,李伟,丁得利,刘春海,姚美玲,朱元芳,王艳菲,李青莹,刘文,苗畅齐,韩永胜. 和牛血液生化指标与肉品质的相关性分析. 畜牧与兽医. 2024(02): 26-30 .  百度学术
百度学术
4. 肖俊,姚远,丁立云,巫伟华,龚海波,傅义龙,陈文静. 饲料蛋白质水平对不同养殖模式下彭泽鲫生长性能、消化酶活性、血液生化指标和抗氧化能力的影响. 饲料工业. 2023(14): 87-92 .  百度学术
百度学术
5. 刘巧凌,黄勇,佟力. 鳜鱼配合饲料养殖试验总结. 河南水产. 2023(03): 18-19 .  百度学术
百度学术
其他类型引用(5)



 下载:
下载: