NEW HOST RECORD AND MOLECULAR PHYLOGENY OF MYXIDIUM SPINIBARBA CHEN ET AL., 2020
-
摘要: 为弄清倒刺鲃两极虫(Myxidium spinibarba)的宿主多样性和胭脂鱼(Myxocyprinus asiaticus)寄生黏孢子虫的种类组成, 研究基于形态和分子数据, 比较分析了寄生于不同宿主的倒刺鲃两极虫的形态学和形态计量学特征及分子系统发育关系。结果显示: 寄生于胭脂鱼和中华倒刺鲃的倒刺鲃两极虫株系在形态学和形态计量学上未出现显著性差异, 18S rDNA序列相似度为99.9%—100.0%, 遗传距离为0.000—0.001, 符合种内变异; 寄生于不同宿主倒刺鲃两极虫的株系在系统发育树中嵌合聚支, 且寄生于胭脂鱼的倒刺鲃两极虫株系先分化。以上结果表明: 研究中两株系与倒刺鲃两极虫为同一物种, 但在分子水平已经出现分化; 这是首次在胭脂鱼中检获到黏孢子虫, 胭脂鱼是倒刺鲃两极虫的新宿主。Abstract: Myxosporeans are microscopic and simple in morphology, mainly parasitize in fish. Some species can cause host disease even lead to death. Among them, the species of the family Myxidiidae Thélohan, 1892 exhibit the broadest range of hosts among all myxosporeans. They are typically coelozoic (rarely histozoic) parasites of marine and freshwater fishes (poikilothermal animals), and a few species are found in another poikilothermal animals (amphibians and reptiles) and homothermal animals (birds and mammals). The genus Myxidium Bütschli, 1882 with 230 described species, currently includes the most known species in the family Myxidiidae. The members of the genus Myxidium exhibit the following characteristics: their myxospores are spindle-shaped, straight, crescent, or even sigmoid with somewhat pointed ends; two pyriform polar capsules are located at both ends of the myxospores; the shell valves are smooth or striated; and the suture line bisects the myxospores. Myxidium spinibarba Chen et al., 2020 was first collected and described from the gall bladder of Spinibarbus sinensis Bleeker, 1871. Present study reported M. spinibarba again from Myxocyprinus asiaticus Bleeker, 1864. Myxocyprinus asiaticus belongs to the family Catostomidaeis, in which only one species has been reported from China. Myxocyprinus asiaticus is a kind of omnivorous benthic fish with important ornamental and edible value. To improve higher economic benefits, M. asiaticus and other fishes with different diets and habitat preferences are often mixed feeding. However, polyculture creates conditions for material exchanges between M. asiaticus and other fishes, and, it also enhances the transmission of parasites or pathogens. To study the host diversity for M. spinibarba and the species composition of myxosporean from M. asiaticus, we analyzed the morphometry characteristics and phylogenetic relationships of M. spinibarba from different hosts based on the morphological and molecular data. Myxosporeans were isolated and photographed from the gallbladder of different M. asiaticus in the same batch. Genomic DNA were extracted and 18S rDNA was amplified and sequenced. Myxospores were fusiform in frontal view, (12.6±0.6) μm (11.6—13.4 μm) long, (5.9±0.4) μm (5.1—6.9 μm) wide, and possessed distinctly longitudinal ridges. The two polar capsules were pear-shaped at the ends of the spore, with a length in (3.48±0.36) μm (2.63—4.22 μm) and a width of (2.83±0.26) μm (2.29—3.21 μm) with 4—5 turns polar filaments. The distance between two polar capsules was (3.66±0.29) μm (3.12—4.20 μm), and their openings were inclined towards the spore ends. These two sequences have the highest similarity with M. spinibarba (MH766654) parasitizing in S. sinensis (99.9%—100.0%). They shared 97.4%—97.7% identity with Zschokkella sp. (KM401441) from Labeo rohita Hamilton, 1822 and Myxidium sp. (MK913426) from Cranoglanis bouderius Richardson, 1846, respectively. Phylogenetic trees of the selected sequences were divided into branch A and B. Clade A was composed Myxidium and Zschokkella species from marine and freshwater fishes or amphibians. All species in clade B were collected from marine fish. Two strains in this study were located in branch A, where the strain (MT775466) and M. spinibarba (MH766654) were first clustered and then grouped with the strain (MT775467). The results of PCA showed that the two strains of this study (MT775466 and MT775467) and M. spinibarba (MH766654) overlapped on the scatter plot. The similarities and genetic distances of 18S rDNA sequences for three strains were 99.9%—100.0% and 0.000—0.001, respectively. Therefore, we conclude that these three strains from different hosts are the same species as M. spinibarba, with diverged molecular level, and that this is the first report of myxosporean in M. asiaticus.
-
Keywords:
- Myxidium spinibarba /
- Myxocyprinus asiaticus /
- New host record /
- Molecular phylogeny /
- 18S rDNA
-
黏孢子虫是一类个体微小结构简单的后生动物寄生虫, 主要寄生于鱼类, 有些种类能引起鱼类宿主的病害[1-3], 严重的甚至会导致死亡[4-7]。其中两极虫科(Myxidiidae Thélohan), 1892黏孢子虫与大多数黏孢子虫类群不同, 具有最广泛的宿主类群, 该科的种类不仅能寄生于变温动物(海、淡水鱼类、两栖类、爬行类)[8-12], 还能寄生于恒温动物(鸟类、哺乳类)[13, 14]。有些物种还能广泛寄生于多种不同的宿主, 如李氏肠黏虫(Enteromyxum leei Diamant, Lom and Dykova), 1994的宿主已经超过40种[15]。两极虫属(Myxidium Butschli, 1882)隶属黏体门(Myxozoa)、黏孢子虫纲(Myxosporea)、双壳目(Bivalvulida)、两极虫科(Myxidiidae)[16], 是两极虫科中种类最多的属, 迄今已记录的种类超过230种[9]。两极虫属种类具如下特征: 2个孢子壳瓣, 孢子呈纺锤形、梨形或椭圆形, 末端稍尖; 壳瓣表面光滑或具条纹, 孢子缝线直; 孢子的两极端各具一个梨形或球形的极囊且极囊向两端开口, 细长的极丝在极囊内横向盘曲; 孢子质位于两极囊之间。两极虫为典型的腔寄生类群, 绝大多数种类寄生于宿主的胆囊或泌尿系统, 极少数可营组织寄生[2, 17], 个别组织寄生种类甚至能导致寄生部位出现空泡影响宿主发育和繁殖[18]。
胭脂鱼(Myxocyprinus asiaticus Bleeker, 1864), 隶属鲤形目(Cypriniformes)胭脂鱼科(Catostomidae)胭脂鱼属(Myxocyprinus), 是胭脂鱼科唯一分布在我国的特有种[19, 20], 现已在我国驯化为养殖鱼类, 成为我国长江流域重要经济鱼类之一[21, 22]。胭脂鱼是杂食性的底栖鱼类, 在人工养殖过程中常与其他不同食性和不同水层栖息的鱼类混养以充分利用不同水层的资源[23]。但混养易增加胭脂鱼与其他不同鱼类的密切接触机会, 加大寄生虫或其他病原传播的风险, 进而增高传染性造成经济损失[24]。因此开展对胭脂鱼的寄生虫及其病害的研究也是水产养殖中鱼病防治的需要。
倒刺鲃两极虫(Myxidium spinibarba Chen, et al.), 2020年最初从中华倒刺鲃[Spinibarbus sinensis (Bleeker, 1871)] 的胆囊中检获[25]。本研究检获的倒刺鲃两极虫分离自胭脂鱼胆囊, 在对其重描述的基础上, 结合其形态学与18S rDNA分子数据, 比较分析寄生于不同宿主的倒刺鲃两极虫及其他相似种之间的分子系统学关系, 旨在为寄生胭脂鱼的黏孢子虫种类组成及两极虫的宿主多样性积累基础数据。
1. 材料与方法
1.1 黏孢子虫样本采集和鉴定
宿主鱼于2019年10月11日从重庆北碚区某养殖基地(该养殖基地由8个8.35 m×5.66 m的养殖池相连, 且池水互通; 多种鱼类以不同年龄、不同规格同池或者单独轮养轮放)捕获。共获26尾无明显病症的胭脂鱼带回实验室解剖并进行黏孢子虫检测, 其中4尾鱼的胆囊检获到黏孢子虫(感染率为15.4%)。黏孢子虫收集、处理、测量和鉴定方法参照文献[26]处理。
1.2 DNA提取、PCR扩增和测序
从上述同一批次不同胭脂鱼个体的胆囊内分离获得游离黏孢子虫, 加入95%的乙醇溶液置于1.5 mL离心管保存。从离心管底部吸取10 μL黏孢子虫液体, 用超纯水清洗2—3次, 离心(2000 ×g)沉淀后用于DNA提取。基因组DNA的提取严格参照DNeasy Blood & Tissue试剂盒(QIAGEN, Hilden, Germany)说明书执行, 将提取的基因组DNA置于–20℃保存、备用。黏孢子虫18S rDNA序列通过PCR技术利用引物18e: 5′-CTGGTTGATCCTGCC-3′[27]和18R: 5′-CTACGGAAACCTTGTTACG-3′[28]扩增。反应体系和反应条件参照文献[29]。取3 μL扩增产物用于1%琼脂糖凝胶进行电泳检测, 目的产物经胶回收试剂盒(OMEGA, 美国)回收并送到上海英潍捷基贸易有限公司测序。
1.3 系统发育分析
将本研究获得的序列, 在GenBank中用BLAST进行同源序列检索比对。根据比对结果, 选取12条高同源性序列(序列相似度高于89%), 24条先前相关研究所用序列[25]及本研究所获的2条序列进行系统发育分析。另以Tetracapsuloides bryosalmonae (KF731712)和Buddenbrockia plumatellae(KF731691)为外群, 利用Mr Bayes 3.2.6[30]选用最优拟合模型(GTR)运行1000000代构建Bayes(BI)树, 利用在线软件CIPRES Science Gateway V.3.3(http://www.phylo.org/)选用RAxML-HPC2 XSEDE(8.2.12)模型构建ML(Maximum likelihood)树。最后用FigTree v1.4和Adobe Photoshop CC软件编辑注释系统树。将相关18S rDNA序列用MEGA 6.0[31]设置执行K2P模型多序列比对计算出遗传距离, 利用在线序列比对工具(http://www.ebi.ac.uk/Tools/psa/)计算得出两两序列相似度, 结合MEGA和Bioedit软件完成变异位点分析。
1.4 主成分分析
使用PAST 3软件进行主成分分析(Principal Component Analysis, PCA), 对不同宿主寄生的倒刺鲃两极虫株系的孢子长、孢子宽、极囊长、极囊宽和极囊间距5项形态学特征进行形态计量学比较[32]。利用变异协方差矩阵生成置信度为95%的散射图。
2. 结果
2.1 倒刺鲃两极虫的形态学重描述
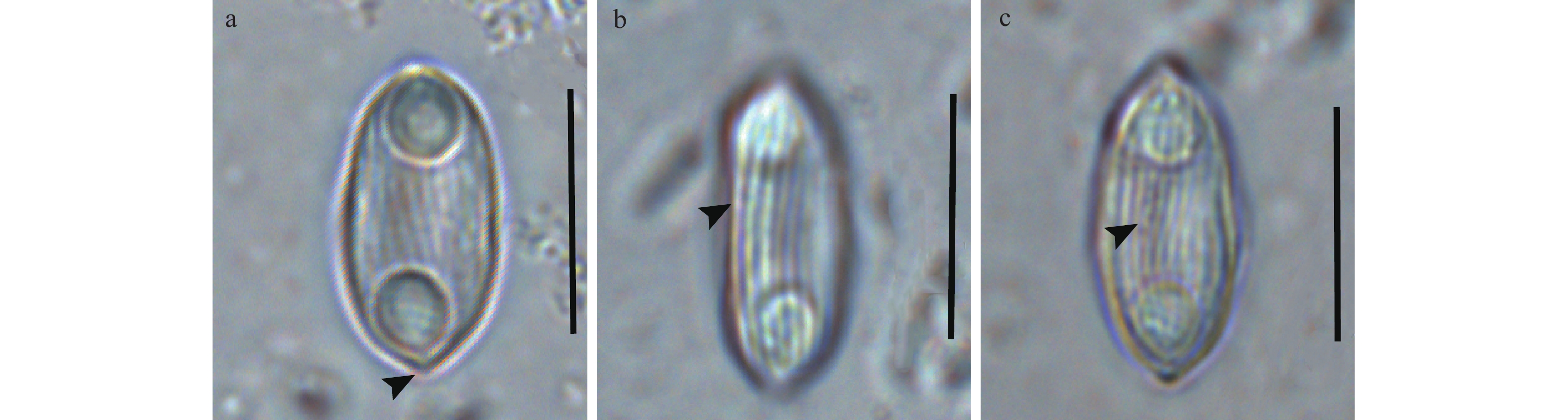
取宿主鱼的适量胆汁制片镜检, 未见营养体和未成熟孢子, 但可见大量成熟孢子。成熟孢子壳面呈梭形, 两端稍尖。孢子缝面呈卵圆形, 缝脊平直, 孢子壳瓣上有7—10条与缝脊平行的条纹(图 1)。孢子长为(12.59±0.56) μm (11.55—13.36 μm), 孢子宽为(5.92±0.40) μm (5.11—6.86 μm)。两个极囊呈梨形位于孢子两端, 大小相等, 其开口倾向孢子两端。极囊长为(3.48±0.36) μm (2.63—4.22 μm), 极囊宽为(2.83±0.26) μm (2.29—3.21 μm)。两极囊间隔(3.66±0.29) μm (3.12—4.20 μm), 极丝盘曲4—5圈(表 1)。
![]() 图 1 寄生于胭脂鱼的倒刺鲃两极虫孢子显微图片a. 孢子壳面观(箭头示孢子尖端); b. 孢子斜缝面观(箭头示孢子缝线); c. 孢子壳面观(箭头示孢子壳面条纹); 比例尺=10 μmFigure 1. Photomicrographs of Myxidium spinibarba from Myxocyprinus asiaticusa. Spore from valvular view (arrowhead showing protrusive poles); b. Spore from slant sutural view (arrowhead showing sutural line); c. Spore from valvular view (arrowhead showing spore striations). Scale bar=10 μm表 1 寄生不同宿主的倒刺鲃两极虫的特征比较Table 1. Comparison among Myxidium spinibarba from different hosts
图 1 寄生于胭脂鱼的倒刺鲃两极虫孢子显微图片a. 孢子壳面观(箭头示孢子尖端); b. 孢子斜缝面观(箭头示孢子缝线); c. 孢子壳面观(箭头示孢子壳面条纹); 比例尺=10 μmFigure 1. Photomicrographs of Myxidium spinibarba from Myxocyprinus asiaticusa. Spore from valvular view (arrowhead showing protrusive poles); b. Spore from slant sutural view (arrowhead showing sutural line); c. Spore from valvular view (arrowhead showing spore striations). Scale bar=10 μm表 1 寄生不同宿主的倒刺鲃两极虫的特征比较Table 1. Comparison among Myxidium spinibarba from different hosts株系(登录号) Strains
(Accession No.)倒刺鲃两极虫
Myxidium spinibarba (MT775466)倒刺鲃两极虫
Myxidium spinibarba (MT775467)倒刺鲃两极虫
Myxidium spinibarba (MH766654)18S rDNA序列长度18S rDNA sequence length (nt) 1785 1913 1949 宿主Host 胭脂鱼Myxocyprinus asiaticus 胭脂鱼Myxocyprinus asiaticus 中华倒刺鲃Spinibarbus sinensis 寄生部位Infection site 胆囊Gall bladder 胆囊Gall bladder 胆囊Gall bladder 孢子长Spore length (μm) 12.6±0.56 (11.6—13.4) 13.0±0.39 (12.3—13.6) 11.8±0.5 (10.6—12.4) 孢子宽Spore width (μm) 5.9±0.40 (5.1—6.9) 5.7±0.45 (5.0—6.3) 6.1±0.5 (5.5—7.2) 极囊长Polar capsule length (μm) 3.5±0.36 (2.6—4.2) 3.5±0.30 (2.7—4.0) 3.6±0.4 (3.0—4.4) 极囊宽Polar capsule width (μm) 2.8±0.26 (2.3—3.2) 2.9±0.22 (2.3—3.4) 3.0±0.2 (2.7—3.2) 极囊间距 Space between polar capsule (μm) 3.7±0.29 (3.1—4.2) 3.9±0.36 (3.2—4.7) 3.6±0.5 (2.8—4.2) 极丝圈数Polar filament number (μm) 4—5 4—5 4—5 数据来源Reference 本研究Present study 本研究Present study Chen等, 2020[25] Chen et al., 2020 2.2 18S rDNA分子特征和系统发育分析
本研究从不同个体的胭脂鱼样本中获得2条18S rDNA序列, 长度分别为1785(MT775466)和1913 nt (MT775467)。比对结果显示, 所获两条18S rDNA序列之间相似度为100.00%, 在可比对长度(1785 nt)上无变异位点。两条序列在NCBI中BLAST分析显示: 与寄生于中华倒刺鲃的倒刺鲃两极虫(MH766654)的相似度最高, 为99.9%—100.0%, MT775467与Myxidium spinibarba (MH766654)在可比对长度范围内(1913 nt)仅有1个变异位点; 其次与Myxidium sp.(MK913426)和Zschokkella sp.(KM401441)具有较高相似度(表 2)。
表 2 黏孢子虫基于18S rDNA序列遗传距离与相似度Table 2. Genetic distances and similarities of myxosporeans based on 18S rDNA sequences物种名
SpeciesGenBank
登录号
GenBank
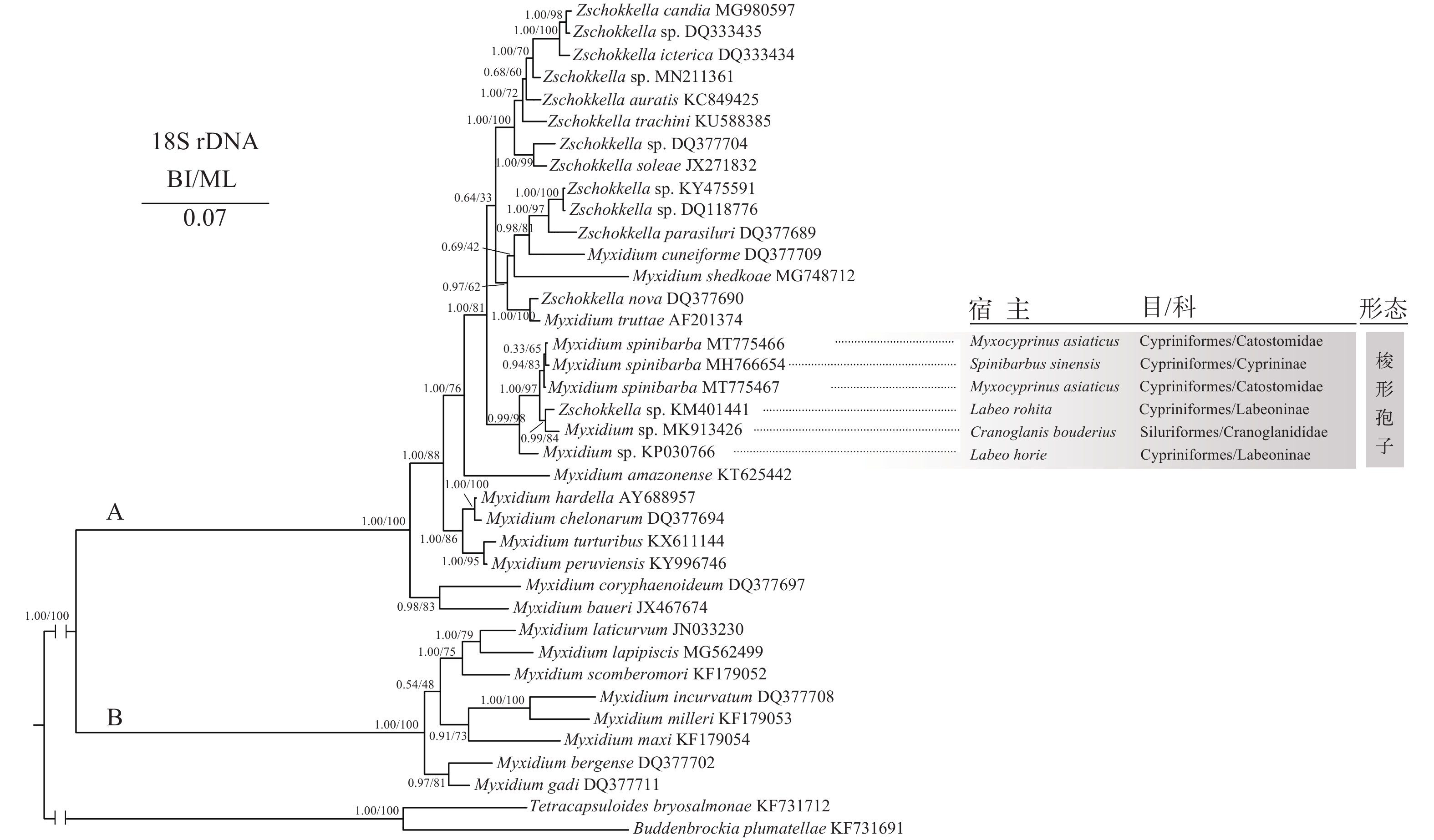
accession No.1 2 3 4 5 1. Myxidium spinibarba MT775466 99.9% 100.0% 97.7% 97.4% 2. Myxidium spinibarba MT775467 0.001 99.9% 97.6% 97.4% 3. Myxidium spinibarba MH766654 0.000 0.001 97.7% 97.5% 4. Myxidium sp. MK913426 0.021 0.022 0.021 97.2% 5. Zschokkella sp. KM401441 0.022 0.023 0.022 0.028 基于18S rDNA序列构建的ML和BI树显示出一致的拓扑结构。两棵系统树均显示: 所选取黏孢子虫序列被分成A、B两个大分支(图 2)。A支是一混合支系, 包括寄生于爬行动物的两极虫、海水鱼类的两极虫和楚克拉虫及淡水鱼类的两极虫和楚克拉虫; 而B支则完全由寄生海水鱼类的两极虫组成。本研究株系位于A支内, 其中株系(MT775466)与倒刺鲃两极虫(MH766654)聚支后, 再与株系(MT775467)聚支, 形成倒刺鲃两极虫支系; 然后, 与Zschokkella sp. (KM401441)和Myxidium sp. (MK913426)亚支再形成了姐妹支。
2.3 基于寄生不同宿主的倒刺鲃两极虫形态学数据的主成分分析
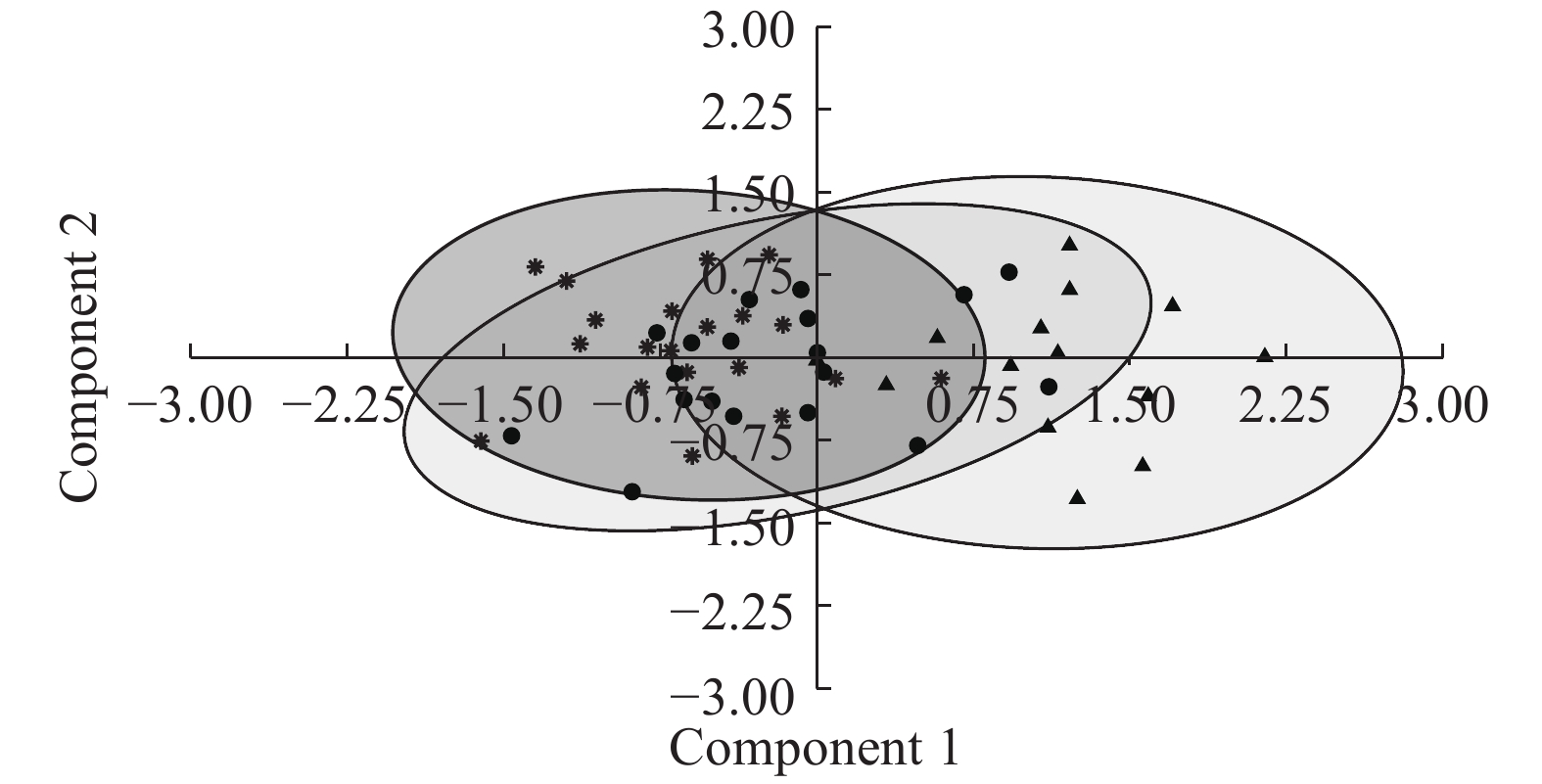
主成分分析结果显示本研究两株系(MT775466和MT775467)与倒刺鲃两极虫(MH766654; 该株系原始数据来自本实验室)在5项形态特征结合生成的椭圆散点图上均有重合, 其中●株系(MT775466)散点图基本覆盖*株系(MH766654)散点图, ▲株系(MT775467)的散点图仅与前两者的散点图部分重叠(图 3)。
![]() 图 3 寄生不同宿主倒刺鲃两极虫的孢子形态差异主成分分析* 倒刺鲃两极虫(MH766654); ● 倒刺鲃两极虫(MT775466); ▲ 倒刺鲃两极虫(MT775467)Figure 3. Principal component analysis on morphological differences of Myxidium spinibarba from different hosts* Myxidium spinibarba (MH766654); ● Myxidium spinibarba (MT775466); ▲ Myxidium spinibarba (MT775467)
图 3 寄生不同宿主倒刺鲃两极虫的孢子形态差异主成分分析* 倒刺鲃两极虫(MH766654); ● 倒刺鲃两极虫(MT775466); ▲ 倒刺鲃两极虫(MT775467)Figure 3. Principal component analysis on morphological differences of Myxidium spinibarba from different hosts* Myxidium spinibarba (MH766654); ● Myxidium spinibarba (MT775466); ▲ Myxidium spinibarba (MT775467)3. 讨论
本研究两株系形态学和生态学特性均显示一致, 应属于同一物种。本研究获得的两极虫, 两端稍尖的梭形成熟孢子、平直的缝脊和壳瓣上的条纹数、两个大小相等的梨形极囊、极丝圈数及极丝开口方向等形态学特征均与倒刺鲃两极虫的形态学特征一致[25], 且两者的感染部位均为胆囊。同时, 形态学数据的主成分分析结果也显示: 尽管株系(MT775467)有略微的差异, 但本研究的2个株系的形态测量数据基本都与倒刺鲃两极虫的形态测量数据有大幅重叠(图 3), 仅存在少许差异, 这样的形态学量度差异在种内变化范围内[33]。综合形态学与主成分分析结果, 本研究获得的两极虫与倒刺鲃两极虫当属同种。
鉴于黏孢子虫结构简单, 学者们在以往的研究中认为在对黏孢子虫进行物种鉴定和描述时, 应当综合考虑该黏孢子虫的形态学特征、生态学特征(宿主、寄生部位、地理分布)和分子特征[34, 35]。基于此, 尽管本研究株系的宿主与原始描述宿主不同, 但其18S rDNA序列比对结果显示寄生胭脂鱼的两株系与中华倒刺鲃株系之间仅有1个变异位点, 遗传距离和相似度分别为0.000—0.001和99.9%—100.0%, 符合已有研究给出的黏孢子虫变异位点不超过10个, 遗传距离在0.000—0.007, 相似度在98.6%—100%属于种内水平的判别标准[36-39]。分子特征分析也表明本研究所获寄生胭脂鱼的两极虫株系(MT77546和MT775467)与寄生中华倒刺鲃的倒刺鲃两极虫(MH766654)为同一物种。
系统发育分析显示: 寄生不同宿主的倒刺鲃两极虫株系嵌合聚枝, 寄生胭脂鱼的倒刺鲃两极虫株系(MT775467)先分化, 并与前述的主成分分析和18S rDNA特征分析结果表现一致。同种两极虫可在亲缘关系较远的鱼类宿主寄生, 这与已有的关于弧形虫和肠黏虫的研究有着明显的一致性[33, 40]。在本研究所显示的倒刺鲃两极虫姐妹支中, 寄生于露斯塔野鲮[Labeo rohita Hamilton, 1822(鲤形目)]的Zschokkella sp.(KM401441)与寄生于长臀[Cranoglanis bouderius Richardson, 1846(鲇形目)]的Myxidium sp.(MK913426)形成高支持率(0.99/84)的系统发生关系也得到了旁证: 尽管Zschokkella sp.(KM401441)与Myxidium sp.(MK913426)为不同种类且宿主亲缘关系较远, 但显示形态学特征较为相似, 都享有梭形的孢子并具有梨形极囊(图 2)。两极虫的系统发生关系与孢子形态、腔寄生的寄生部位趋向性等可能更相关。在通常情况下, 内寄生虫的进化在很大程度上受其与宿主相互作用的影响, 基于黏孢子虫SSU rDNA的大数据集系统发育分析结果认为组织寄生物种是由类似祖先型的腔寄生黏孢子虫进化而来[41, 42]。寄生鱼类的碘泡虫类黏孢子虫多为组织寄生, 是黏孢子虫中进化较晚的类群[42]。绝大多数碘泡虫类的黏孢子虫寄生于淡水鱼且具有明显的宿主特异性乃至寄生部位特异性, 往往只感染一种宿主甚至只感染一种宿主的某一特定器官或组织[35]。而腔寄生的两极虫类是相对组织寄生的碘泡虫类较为祖先的类群, 进化过程中分化形成海水支系和淡水支系, 其变异程度要比碘泡虫等组织寄生类群更大, 更易于对环境及宿主的变化形成适应[41, 42]。此外, 先前基于鱼体混养、受感染鱼宿主的生活水体、经口饲喂和肛门插管等途径对李氏肠黏虫传播实验研究表明, 李氏肠黏虫可感染多种不同且亲缘关系较远的鱼类宿主[43], 也说明腔寄生的两极虫类黏孢子虫对宿主具有更广泛的适应性。鉴于此, 推测本研究中倒刺鲃两极虫也可能具有多宿主特征, 能寄生多种不同宿主, 但这还需要更多研究予以证实。
综上, 本研究首次在胭脂鱼体内检获黏孢子虫, 胭脂鱼是倒刺鲃两极虫的新宿主; 寄生胭脂鱼的倒刺鲃两极虫株系(MT775467)先分化。本研究也为寄生胭脂鱼的黏孢子虫种类组成积累了新的数据。
-
图 1 寄生于胭脂鱼的倒刺鲃两极虫孢子显微图片
a. 孢子壳面观(箭头示孢子尖端); b. 孢子斜缝面观(箭头示孢子缝线); c. 孢子壳面观(箭头示孢子壳面条纹); 比例尺=10 μm
Figure 1. Photomicrographs of Myxidium spinibarba from Myxocyprinus asiaticus
a. Spore from valvular view (arrowhead showing protrusive poles); b. Spore from slant sutural view (arrowhead showing sutural line); c. Spore from valvular view (arrowhead showing spore striations). Scale bar=10 μm
图 3 寄生不同宿主倒刺鲃两极虫的孢子形态差异主成分分析
* 倒刺鲃两极虫(MH766654); ● 倒刺鲃两极虫(MT775466); ▲ 倒刺鲃两极虫(MT775467)
Figure 3. Principal component analysis on morphological differences of Myxidium spinibarba from different hosts
* Myxidium spinibarba (MH766654); ● Myxidium spinibarba (MT775466); ▲ Myxidium spinibarba (MT775467)
表 1 寄生不同宿主的倒刺鲃两极虫的特征比较
Table 1 Comparison among Myxidium spinibarba from different hosts
株系(登录号) Strains
(Accession No.)倒刺鲃两极虫
Myxidium spinibarba (MT775466)倒刺鲃两极虫
Myxidium spinibarba (MT775467)倒刺鲃两极虫
Myxidium spinibarba (MH766654)18S rDNA序列长度18S rDNA sequence length (nt) 1785 1913 1949 宿主Host 胭脂鱼Myxocyprinus asiaticus 胭脂鱼Myxocyprinus asiaticus 中华倒刺鲃Spinibarbus sinensis 寄生部位Infection site 胆囊Gall bladder 胆囊Gall bladder 胆囊Gall bladder 孢子长Spore length (μm) 12.6±0.56 (11.6—13.4) 13.0±0.39 (12.3—13.6) 11.8±0.5 (10.6—12.4) 孢子宽Spore width (μm) 5.9±0.40 (5.1—6.9) 5.7±0.45 (5.0—6.3) 6.1±0.5 (5.5—7.2) 极囊长Polar capsule length (μm) 3.5±0.36 (2.6—4.2) 3.5±0.30 (2.7—4.0) 3.6±0.4 (3.0—4.4) 极囊宽Polar capsule width (μm) 2.8±0.26 (2.3—3.2) 2.9±0.22 (2.3—3.4) 3.0±0.2 (2.7—3.2) 极囊间距 Space between polar capsule (μm) 3.7±0.29 (3.1—4.2) 3.9±0.36 (3.2—4.7) 3.6±0.5 (2.8—4.2) 极丝圈数Polar filament number (μm) 4—5 4—5 4—5 数据来源Reference 本研究Present study 本研究Present study Chen等, 2020[25] Chen et al., 2020 表 2 黏孢子虫基于18S rDNA序列遗传距离与相似度
Table 2 Genetic distances and similarities of myxosporeans based on 18S rDNA sequences
物种名
SpeciesGenBank
登录号
GenBank
accession No.1 2 3 4 5 1. Myxidium spinibarba MT775466 99.9% 100.0% 97.7% 97.4% 2. Myxidium spinibarba MT775467 0.001 99.9% 97.6% 97.4% 3. Myxidium spinibarba MH766654 0.000 0.001 97.7% 97.5% 4. Myxidium sp. MK913426 0.021 0.022 0.021 97.2% 5. Zschokkella sp. KM401441 0.022 0.023 0.022 0.028 -
[1] Kent M L, Andree K B, Bartholomew J L, et al. Recent advances in our knowledge of the Myxozoa [J]. Journal of Eukaryotic Microbiology, 2001, 48(4): 395-413. doi: 10.1111/j.1550-7408.2001.tb00173.x
[2] Lom J, Dyková I. Myxozoan genera: definition and notes on taxonomy, life-cycle terminology and pathogenic species [J]. Folia Parasitologica, 2006, 53(1): 1-36. doi: 10.14411/fp.2006.001
[3] Molnár K. Site preference of myxozoans in the kidneys of Hungarian fishes [J]. Diseases of Aquatic Organisms, 2007, 78(1): 45-53.
[4] Kazuo K. Intestinal giant-cystic disease affecting the carp, caused by Thellohanellus sp. [J]. Fish Pathology, 1980, 14(3): 145-146. doi: 10.3147/jsfp.14.145
[5] Zhai Y H, Gu Z M, Guo Q X, et al. New type of pathogenicity of Thelohanellus kitauei Egusa & Nakajima, 1981 infecting the skin of common carp Cyprinus carpio L [J]. Parasitology International, 2016, 65(1): 78-82. doi: 10.1016/j.parint.2015.10.010
[6] El-Matbouli M, Hoffmann R W, Schoel H, et al. Whirling disease: host specificity and interaction between the actinosporean stage of Myxobolus cerebralis and rainbow trout Oncorhynchus mykiss [J]. Diseases of Aquatic Organisms, 1999, 35(1): 1-12.
[7] Palenzuela O, Redondo M J, Álvarez-Pellitero P. Description of Enteromyxum scophthalmi gen. nov. sp. nov. (Myxozoa), an intestinal parasite of turbot (Scophthalmus maximus L.) using morphological and ribosomal RNA sequence data [J]. Parasitology, 2002, 124(4): 369-379. doi: 10.1017/S0031182001001354
[8] Whipps C, Zhao Y J. Synopsis of the species of the genus Sphaeromyxa Thélohan, 1892 (Myxosporea: Bivalvulida: Variisporina: Sphaeromyxidae) [J]. Systematic Parasitology, 2015, 92(2): 81-99. doi: 10.1007/s11230-015-9591-y
[9] Eiras J C, Saraiva A, Santos M J, et al. Synopsis of the species of Myxidium Bütschli, 1882 (Myxozoa: Myxosporea: Bivalvulida) [J]. Systematics Parasitology, 2011, 80(2): 81-116. doi: 10.1007/s11230-011-9315-x
[10] Hartigan A, Fiala I, Dyková I, et al. New species of Myxosporea from frogs and resurrection of the genus Cystodiscus Lutz, 1889 for species with myxospores in gallbladders of amphibians [J]. Parasitology, 2012, 139(4): 478-496. doi: 10.1017/S0031182011002149
[11] Heiniger H, Adlard R D. Relatedness of novel species of Myxidium Bütschli, 1882, Zschokkella Auerbach, 1910 and Ellipsomyxa Køie, 2003 (Myxosporea: Bivalvulida) from the gall bladders of marine fishes (Teleostei) from Australian waters [J]. Systematic Parasitology, 2014, 87(1): 47-72. doi: 10.1007/s11230-013-9454-3
[12] Espinoza L L, Mertins O, Gama G S, et al. A new Myxidium species (Myxozoa: Myxosporea) infecting the gallbladder of the turtle Podocnemis unifilis (Testudines: Podocnemididae) from Peruvian Amazon [J]. Acta Tropica, 2017(172): 75-79.
[13] Bartholomew J L, Atkinson S D, Hallett S L, et al. Myxozoan parasitism in waterfowl [J]. International Journal for Parasitology, 2008, 38(10): 1199-1207. doi: 10.1016/j.ijpara.2008.01.008
[14] Szekely C, Cech G, Atkinson S D, et al. A novel myxozoan parasite of terrestrial mammals: description of Soricimyxum minuti sp n. (Myxosporea) in pygmy shrew Sorex minutus from Hungary [J]. Folia Parasitologica, 2015, 62(45): 1-5.
[15] Diamant A, Ram S, Paperna I. Experimental transmission of Enteromyxum leei to freshwater fish [J]. Diseases of Aquatic Organisms, 2006, 72(2): 171-178.
[16] Canning E U, Okamura B. Biodiversity and evolution of the Myxozoa [J]. Advances in Parasitology, 2003, 56(2): 43-131.
[17] Fariya N, Kaur H, Abidi R. Myxidium tictoi n. sp. a myxozoan parasite infecting kidney of fresh water barb Puntius ticto (Hamilton, 1822) from river Gomti, Lucknow (U. P) [J]. Journal of Parasitic Diseases, 2019, 44(4): 126-130.
[18] Mohammed A J, Reda H. Ovarian abnormality in a pathological case caused by Myxidium sp. (Myxozoa, Myxosporea) in onespot snapper fish Lutjanus monostigma (Teleostei, Lutjanidae) from the Red Sea [J]. Acta Parasitologica, 2010, 55(1): 1-7. doi: 10.2478/s11686-010-0008-4
[19] 罗云林, 伍献文. 中国胭脂鱼的骨骼形态和胭脂鱼科的分类位置 [J]. 动物分类学报, 1979, 4(3): 195-203. Luo Y L, Wu X W. Anatomical feature of Myxocyprinus asiaticus and its systematic position [J]. Acta Zootaxonomica Sinica, 1979, 4(3): 195-203.
[20] 孙玉华, 刘思阳, 王伟, 等. 中国胭脂鱼线粒体控制区遗传多样性分析 [J]. 遗传学报, 2002, 29(2): 787-790. Sun Y H, Liu S Y, Wang W, et al. Genetics diversity analysis of mitochondrial D-Loop region of Chinese sucker (Myxocyprinus asiaticus) [J]. Acta Genetica Sinica, 2002, 29(2): 787-790.
[21] 张春光, 赵亚辉, 康景贵. 我国胭脂鱼资源现状及其资源恢复途径的探讨 [J]. 自然资源学报, 2000, 15(2): 155-159. doi: 10.3321/j.issn:1000-3037.2000.02.011 Zhang C G, Zhao Y H, Kang J G. A discussion on resources status of Myxocyprinus asiaticus (Bleeker) and their conservation and the recovery [J]. Journal of Natural Resources, 2000, 15(2): 155-159. doi: 10.3321/j.issn:1000-3037.2000.02.011
[22] 吴土金. 胭脂鱼人工驯养与繁育技术研究 [J]. 海洋与渔业, 2016, 263(3): 62-63. Wu T J. Research on artificial domestication and breeding technology of Myxocyprinus asiaticus [J]. Ocean and Fishery, 2016, 263(3): 62-63.
[23] 王桂学, 沈志刚, 钟巧仙. 胭脂鱼专、主养模式养殖对比试验 [J]. 水产养殖, 2012, 33(5): 9-11. doi: 10.3969/j.issn.1004-2091.2012.05.005 Wang G X, Shen Z G, Zhong Q X. Comparison test between intensive and primary aquaculture mode of Myxiocyprinus asiaticus [J]. Journal of Aquaculture, 2012, 33(5): 9-11. doi: 10.3969/j.issn.1004-2091.2012.05.005
[24] 王桂良, 韩光明, 寇祥明, 等. 不同胭脂鱼混养模式的产量与综合效益分析 [J]. 海洋渔业, 2018, 40(2): 197-206. doi: 10.3969/j.issn.1004-2490.2018.02.009 Wang G L, Han G M, Kou X M. Yields and comprehensive benefits of different polyculture modes of Chinese sucker Myxocyprinus asiaticus [J]. Marine Fisheries, 2018, 40(2): 197-206. doi: 10.3969/j.issn.1004-2490.2018.02.009
[25] Chen W, Yang C Z, Zhao Y J. Characterization of Myxidium spinibarba sp. nov. (Cnidaria, Myxosporea, Myxidiidae) from Spinibarbus sinensis (Bleeker, 1871) in Chongqing China [J]. Parasitology Research, 2020, 119(5): 1485-1491. doi: 10.1007/s00436-020-06644-0
[26] 赵元莙, 马成伦, 宋微波. 海水养殖中原生动物病原虫的鉴定: II. 黏孢子虫原生动物的研究及鉴定方法 [J]. 青岛海洋大学学报, 2001, 31(6): 681-688. Zhao Y J, Ma C L, Song W B. Illustrated guide to the identification of pathogenetic Protozoa in mariculture-II. Diagnostic methods for the Myxosporea [J]. Journal of Ocean University Qingdao, 2001, 31(6): 681-688.
[27] Hillis D M, Dixon M T. Ribosomal DNA: molecular evolution and phylogenetic inference [J]. The Quarterly Review of Biology, 1991, 66(4): 411-453. doi: 10.1086/417338
[28] Whipps C M, Adlard R D, Bryant M S, et al. First report of three Kudoa species from Eastern Australia: Kudoa thyrsites from Mahi mahi (Coryphaena hippurus), Kudoa amamiensis and Kudoa minithyrsites n. sp. from Sweeper (Pempheris ypsilychnus) [J]. Journal of Eukaryotic Microbiology, 2003, 50(3): 215-219. doi: 10.1111/j.1550-7408.2003.tb00120.x
[29] Liu X H, Zhang D D, Yang C Z, et al. Morphological and molecular identification of Myxobolus parakoi sp. Nov (Myxozoa: Myxobolidae), from Cyprinus carpio in Chongqing China [J]. Zootaxa, 2019, 4657(1): 117-126. doi: 10.11646/zootaxa.4657.1.4
[30] Ronquist F, Huelsenbeck J P. MrBayes 3: Bayesian phylogenetic inference under mixed models [J]. Bioinformatics, 2003, 19(12): 1572-1574. doi: 10.1093/bioinformatics/btg180
[31] Tamura K, Stecher G, Peterson D, et al. MEGA6: Molecular evolutionary genetics analysis version 6.0 [J]. Molecular Biology and Evolution, 2013, 30(12): 2725-2729. doi: 10.1093/molbev/mst197
[32] Hammer Ø, Harper DAT, Ryan P D. PAST: paleontological statistics software package for education and data analysis [J]. Palaeontol Electron, 2001(4): 1-9.
[33] Chen W, Yang C Z, Whipps C, et al. Taxonomy on three novel species of Sphaeromyxa Thélohan, 1892 (Myxozoa, Bivalvulida, Sphaeromyxidae) with insight into the evolution of the genus [J]. Parasitology Research, 2020, 119(5): 1493-1503. doi: 10.1007/s00436-020-06656-w
[34] Fiala I, Bartošová S P, Whipps C M. Classification and Phylogenetics of Myxozoa [M]. Myxozoan Evolution, Ecology and Development. Springer International Publishing, 2015: 85-110.
[35] Molnár K, Székely C, Hallett S L, et al. Some remarks on the occurrence, host-specificity and validity of Myxobolus rotundus Nemeczek, 1911 (Myxozoa: Myxosporea) [J]. Systematic Parasitology, 2009, 72(1): 71-79. doi: 10.1007/s11230-008-9161-7
[36] Zhao Y J, Li N N, Tang F H. Remarks on the validity of Myxobolus ampullicapsulatus and Myxobolus honghuensis (Myxozoa: Myxosporea) based on SSU rDNA sequences [J]. Parasitology Research, 2013, 112(11): 3817-3823. doi: 10.1007/s00436-013-3569-y
[37] 冉佼, 杨承忠, 赵元莙. 基于遗传距离的黏孢子虫分类研究 [J]. 重庆师范大学学报(自然科学版), 2013, 31(3): 31-34. Ran J, Yang C Z, Zhao Y J. Taxonomic research of the Myxosporidia based on genetic distance [J]. Journal of Chongqing Normal University (
Natural Science Edition ) , 2013, 31(3): 31-34. [38] Karlsbakk E, Kristmundsson Á, Albano M, et al. Redescription and phylogenetic position of Myxobolus aeglefini and Myxobolus platessae n. comb. (Myxosporea), parasites in the cartilage of some North Atlantic marine fishes, with notes on the phylogeny and classification of the Platysporina [J]. Parasitology International, 2017, 66(1): 952-959. doi: 10.1016/j.parint.2016.10.014
[39] 刘晓聪, 杨承忠, 赵元莙. 洪湖碘泡虫的再描述及其近缘种的鉴别性研究 [J]. 水生生物学报, 2016, 40(2): 350-357. Liu X C, Yang C Z, Zhao Y J. Redescription of Myxobolus honghuensis and identification on its genetic related species [J]. Acta Hydrobiologica Sinica, 2016, 40(2): 350-357.
[40] Miller T L, Barnett S K, Seymour J E, et al. Biliary tract-infecting Myxosporeans from estuarine and reef stonefish (Scorpaeniformes: Synanceiidae) off Eastern Australia, with descriptions of Sphaeromyxa horrida n. sp. and Myxidium lapipiscis n. sp. (Myxosporea: Bivalvulida) [J]. Journal of Parasitology, 2018, 104(3): 254-261. doi: 10.1645/17-79
[41] Fiala I. The phylogeny of Myxosporea (Myxozoa) based on small subunit ribosomal RNA gene analysis [J]. International Journal for Parasitology, 2006, 36(14): 1521-1534. doi: 10.1016/j.ijpara.2006.06.016
[42] Fiala I, Bartošová P. History of myxozoan character evolution on the basis of rDNA and EF-2 data [J]. BMC Evolutionary Biology, 2010, 10(1): 228-240. doi: 10.1186/1471-2148-10-228
[43] Estensoro I, JosRedondo M, Alvarez-Pellitero P, et al. Novel horizontal transmission route for Enteromyxum leei (Myxozoa) by anal intubation of gilthead sea bream Sparus aurata [J]. Diseases of Aquatic Organisms, 2010, 92(1): 51-58. doi: 10.3354/dao02267



 下载:
下载: